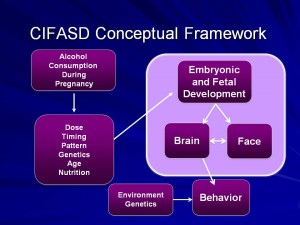
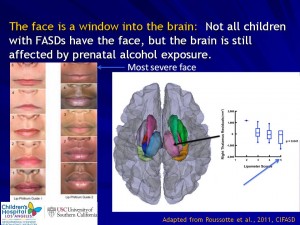
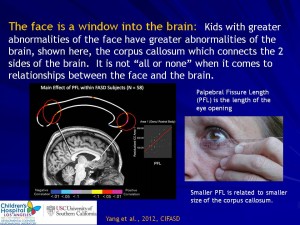
Research Projects and Components
- Administration of CIFASD (Edward P. Riley, PI)
- Prenatal alcohol effects on physical development (Kenneth L. Jones, PI)
- Identification of the facial features of FASD with 3D imaging (Tatiana Foroud & Peter Hammond, PIs)
- Prenatal alcohol effects on brain and facial development (Kathleen K. Sulik, PI)
- Prenatal alcohol effects on brain development and how it relates to behavior (Elizabeth Sowell, PI)
- Prenatal alcohol effects on behavior and cognition (Sarah Mattson, PI)
- Early identification of FASD and nutrition (Christina Chambers, PI)
- CIFASD data management (William K. Barnett, PI)
- Genes and FASD (Johann Eberhart, PI)
- FASD, stress and daily rhythms (Dipak Sarkar, PI)
- Prenatal alcohol effects on early brain development (Kirsty Donald, PI)
- Prenatal alcohol effects on immune function (Joanne Weinberg, PI)
Administration of CIFASD
 The more we know about what happens to the developing baby when a pregnant woman drinks alcohol, the better we are able to treat and prevent fetal alcohol spectrum disorders (FASD). This project brings together people who study FASD with various methods and helps them collaborate to better understand FASD. For example, can we improve our ability to identify a person who has been exposed to alcohol during development? To reach our goals, we arrange regular meetings so that each research group knows what the other research groups are doing. We help them share resources and data. New projects are identified and assisted, which we hope will help us solve the problems of FASD faster. Our goal is to end FASD, and to help find better treatments for those who are affected.
The more we know about what happens to the developing baby when a pregnant woman drinks alcohol, the better we are able to treat and prevent fetal alcohol spectrum disorders (FASD). This project brings together people who study FASD with various methods and helps them collaborate to better understand FASD. For example, can we improve our ability to identify a person who has been exposed to alcohol during development? To reach our goals, we arrange regular meetings so that each research group knows what the other research groups are doing. We help them share resources and data. New projects are identified and assisted, which we hope will help us solve the problems of FASD faster. Our goal is to end FASD, and to help find better treatments for those who are affected.
Edward P. Riley, Principal Investigator
Administrative Core of the CIFASD U24AA014811-10 | San Diego State University
The Administrative Core (AdminC) of the Collaborative Initiative on Fetal Alcohol Spectrum Disorders (CIFASD) oversees a multidisciplinary, multisite consortium consisting of both basic science and clinical components, with the aim of improving the diagnostic criteria for the range of outcomes following prenatal alcohol exposure. The consortium consists of three cores (AdminC, Dysmorphology, and Informatics), clinical U01s, and basic science U01s. These projects are structured to be collaborative and interrelated. The AdminC maintains the organizational framework for the CIFASD, managing and supporting the two components and their Steering Committees, and providing support to the Science Advisory Board. Besides its administrative functions, the AdminC is also responsible for two additional components, the Developmental and Educational Components of the CIFASD. The Developmental Component provides a mechanism for the inclusion of new research ideas, potential new sites, and higher risk/higher payoff projects. The Educational Component provides a new mechanism to ensure that consortium findings are widely circulated to the public and other stakeholders in a timely fashion. The Specific Aims of the AdminC are to: 1) Provide scientific and administrative leadership and oversight, ensuring that CIFASD investigators adhere to its goals and mission, as well as assisting and coordinating interactions between the various projects. Function as the main liaison to NIAAA regarding the CIFASD; 2) Facilitate communication between the various projects by maintaining the CIFASD web site, scheduling monthly conference calls, convening the biannual meetings of the Steering Committees, and preparing and distributing annual progress reports; 3) Provide assistance with data collection and distribution by working with the Informatics Core. Work with the Informatics Core to develop online interactive capacity among CIFASD investigators, and make most of these databases available to outside, interested scientists; 4) Assist the Science Advisory Board and the Steering Committees in their annual evaluations of progress for each of the projects. In conjunction with the Steering Committees, it will establish annual priorities and address issues related to allocation of resources; and 5) Administer the Developmental and Educational Components of the CIFASD.
PUBLIC HEALTH RELEVANCE: The consequences resulting from prenatal alcohol exposure are wide-ranging and are major public health issues. The goal of the research proposed by the CIFASD is to enhance the diagnosis of fetal alcohol spectrum disorders across the range of outcomes, using a multisite, multidisciplinary consortium consisting of both basic science and clinical projects.
Prenatal alcohol effects on physical development
 Many things can affect development of a child, including prenatal exposure to alcohol. Although there are no lab tests that can diagnosis FASD, there are a number of subtle physical features that may help us identify kids with FASD. Our group is creating a DVD that will be available to physicians and other healthcare providers that shows how one can examine and identify these subtle features. In addition, we are creating a telemedicine system that will allow experts to help healthcare providers in areas throughout the world who do not have the experience to diagnose FASD. We recognize that getting a diagnosis of FASD is often a major challenge for families. Given the subtlety of the physical features, many physicians do not yet have the necessary skills, which can be frustrating for parents. Our goal is to develop ways to improve diagnosis and help these families.
Many things can affect development of a child, including prenatal exposure to alcohol. Although there are no lab tests that can diagnosis FASD, there are a number of subtle physical features that may help us identify kids with FASD. Our group is creating a DVD that will be available to physicians and other healthcare providers that shows how one can examine and identify these subtle features. In addition, we are creating a telemedicine system that will allow experts to help healthcare providers in areas throughout the world who do not have the experience to diagnose FASD. We recognize that getting a diagnosis of FASD is often a major challenge for families. Given the subtlety of the physical features, many physicians do not yet have the necessary skills, which can be frustrating for parents. Our goal is to develop ways to improve diagnosis and help these families.
Kenneth L. Jones
Dysmorphology Core U24AA014815-10 | University of California San Diego
Although the brain is the developing structure most sensitive to alcohol, the diagnosis of Fetal Alcohol syndrome (FAS), which represents the most severe end of the spectrum of Fetal Alcohol Spectrum Disorders (FASD), is presently dependent, not upon alterations of brain structure or behavior, but upon features identified on a careful physical examination. Over the previous funding periods the Dysmorphology Core has evaluated 345 subjects with FAS, 771 who have been Deferred and 502 with NO FAS at consortium sites throughout the world. The specific aims of this proposal are to continue to support these activities throughout the renewal period at all consortium sites, to develop a training DVD that could be used to teach physicians and other health care professionals with little or no experience in diagnosis of FASD to successfully diagnose or rule out this disorder, to develop with the help of the Telemedicine Communications Center at the University of California, San Diego (UCSD), a wireless, interactive, audiovisual tele-consultation system that will provide the opportunity for a practitioner in a remote area to perform a comprehensive examination of a child considered to have Fetal Alcohol Spectrum Disorders (FASD) that could be simultaneously viewed, by an expert dysmorphologist at UCSD, and to document the prevalence of major malformations in children prenatally exposed to alcohol, and in so doing, delineate the extent of Alcohol Related Birth Defects (ARBD). Furthermore, the development of a training DVD to teach physicians and other health care providers an approach to diagnosis of FASD will help fill the gap in access to expert diagnosticians. Finally, given the significant morbidity and mortality associated with major structural defects, a rigorous evaluation for Alcohol Related Birth Defects (ARBD) will give important information regarding the need to screen children for these defects based on having been exposed prenatally to alcohol.
PUBLIC HEALTH RELEVANCE: Relative to the relevance of this research to public health, through collaboration with other CIFASD projects, we will to delineate the full spectrum of defects that are associated with prenatal alcohol exposure. Only by accomplishing this will it be possible to determine the true incidence of problems that alcohol imposes on the developing human fetus, a requirement for developing and carrying out programs to prevent it.
Identification of the facial features of FASD with 3D imaging

 We are using a photograph of the face to improve our ability to identify people exposed to alcohol prenatally. We take pictures of faces from children in the U.S., South Africa and the Ukraine. In addition to children with prenatal alcohol exposure and children not exposed to alcohol, we look at the faces of children with other developmental problems. Our goal is to better understand how the face changes. The study will help us understand the effects of prenatal alcohol exposure on the face and determine which features are specific to the effects of prenatal alcohol and which are shared with other conditions. Our study will also help us identify individuals with prenatal alcohol exposure at a younger age and provide a tool to screen children in the clinic. Early identification of fetal alcohol exposure could help us treat the individual and improve outcome.
We are using a photograph of the face to improve our ability to identify people exposed to alcohol prenatally. We take pictures of faces from children in the U.S., South Africa and the Ukraine. In addition to children with prenatal alcohol exposure and children not exposed to alcohol, we look at the faces of children with other developmental problems. Our goal is to better understand how the face changes. The study will help us understand the effects of prenatal alcohol exposure on the face and determine which features are specific to the effects of prenatal alcohol and which are shared with other conditions. Our study will also help us identify individuals with prenatal alcohol exposure at a younger age and provide a tool to screen children in the clinic. Early identification of fetal alcohol exposure could help us treat the individual and improve outcome.
Tatiana Foroud and Peter Hammond
3D Facial Imaging in FASD U01AA014809-10 | Indiana Univ. – Purdue Univ. at Indianapolis | University College London
This project will focus on several of the specific research areas targeted in the Request for Application (RFA) including: a) improvements in the diagnosis of FASD, b) enhanced understanding of FASD dysmorphology through 2-D and 3-D image analysis, and c) earlier case identification. We will work collaboratively with each of the clinical projects recruiting the subjects from whom we will obtain facial images. In addition, we will compare results with the two basic science projects that are focusing on the use of animal models. Lastly, we will rely on the two cores for subject evaluation (Dysmorphology) and data management (Bioinformatics). Throughout this application and within the Consortium – there is a strong focus on novel methods to better understand and recognize the spectrum of deficits resulting from prenatal alcohol exposure. Furthermore, replication of results is a key component of our study design. We seek not only to replicate key findings within the same research population – but to also extend the results to other populations of differing ethnicity and diversity. Such worldwide replication is only possible through an international consortium like CIFASD. Results from this study will then be used as the basis for new screening tools that can be used by clinicians. To accomplish our goals, we propose the following specific aims: 1) Develop a screening tool that will utilize the data from the 3D facial images and support accurate identification of individuals with a high likelihood of alcohol exposure. 2) Recruit and analyze facial imaging data from very young populations to develop a screening tool that accurately identifies high risk individuals for future intervention. 3) Combine face images, neurobehavioral data and brain images to identify common pathways and hence improve diagnosis of prenatal alcohol exposure. 4) Extend existing and develop novel techniques and associated software to cope with demands of larger datasets and more diverse comparison of controls, alcohol exposed and other developmentally delayed subjects while accommodating multiple anatomical images per subject 5) Extend preliminary genetic studies through collection of DNA samples for new subjects and focused analysis to replicate candidate genes identified in basic science components.
PUBLIC HEALTH RELEVANCE: The goal of this project and the Consortium as a whole is to develop new methods to better understand and recognize the spectrum of deficits that result from prenatal alcohol exposure. This project is focused on the use of facial images as the primary approach to this question. We will use facial images to improve the diagnosis of fetal alcohol spectrum disorders and also develop methods to identify these children at very early ages – so as to allow early intervention to improve outcome.
Prenatal alcohol effects on brain and facial development
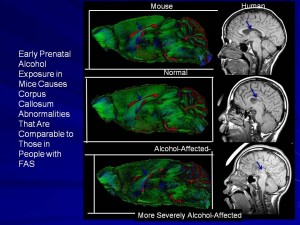
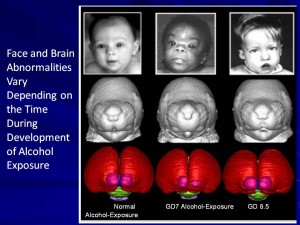
 We use a mouse model of FASD to study the effects of alcohol on the developing face and brain. Unlike human studies, work with animals allows careful control over important factors such as exposure time during development and amount of alcohol. We use sophisticated methods to image the face and the brain of animals exposed to alcohol prenatally. This helps us recognize how subtle changes in the face may tell us about the effects of alcohol on the brain. Importantly, our work has shown that alcohol can damage the face and brain when exposure occurs at times prior to when a woman usually knows she is pregnant. This has major implications for prevention efforts. If there is a chance that a woman might become pregnant, she should avoid alcohol.
We use a mouse model of FASD to study the effects of alcohol on the developing face and brain. Unlike human studies, work with animals allows careful control over important factors such as exposure time during development and amount of alcohol. We use sophisticated methods to image the face and the brain of animals exposed to alcohol prenatally. This helps us recognize how subtle changes in the face may tell us about the effects of alcohol on the brain. Importantly, our work has shown that alcohol can damage the face and brain when exposure occurs at times prior to when a woman usually knows she is pregnant. This has major implications for prevention efforts. If there is a chance that a woman might become pregnant, she should avoid alcohol.
Kathleen K. Sulik
Craniofacial and CNS Pathology in a Mouse FASD Model U01AA021651-02 | University of North Carolina Chapel Hill
The objective of our basic research is to make clinically-relevant discoveries regarding prenatal alcohol (ethanol) exposure-induced pathology involving the brain and face. This work builds naturally on our CIFASD-supported basic research to date and continues to address the need for a more complete understanding of the spectrum and exposure stage-dependency of abnormalities caused by maternal alcohol use. Utilizing a well-established FASD mouse model, along with innovative technologies and approaches, and addressing 3 Specific Aims, we are testing the overall hypothesis that alcohol induces structural abnormalities of the brain and face in mice that are consistent with and informative for those in human FASD. The Aim 1 studies compliment and extend the clinical research of Drs. Foroud and Hammond. They employ Magnetic Resonance Imaging (MRI) and dense surface modeling (DSM) for experiments that are designed to identify exposure stage-dependent correlative abnormalities of the brain and face. The Aim 2 studies compliment and extend the Sowell group’s neuroimaging-based clinical studies while following up on preliminary findings of cerebro-cortical thickness alterations in our mouse model. For this, MRI-based assessments of regional brain volumes and cerebro-cortical thickness changes, along with DTI-based investigations of fiber tract and structural connectivity alterations in adult animals are being conducted. The Aim 3 studies are directed toward further defining the histopathology and genesis of early prenatal alcohol exposure-induced regional brain dysmorphology. They employ routine histological methods, as well as immunohistochemistry, and stereology. Specimens selected for detailed histological analyses include those postnatal brains that had previously been imaged and analyzed for Aim 2. Additional Aim 3 studies focus on prenatal stages and address the novel concept that early alcohol insult yields changes in Cajal-Retzius cell populations; changes that underlie subsequent cerebro-cortical lamination defects. The results of these studies promise to facilitate diagnosis of the full range of birth defects associated with prenatal alcohol exposure and will aid in elucidating biological mechanisms that contribute to alcohol teratogenesis. They also promise to fill a significant FASD research void, inform human clinical research, and continue to highlight the first trimester as a critical period for alcohol-induced defects of the face and brain.
PUBLIC HEALTH RELEVANCE: Fetal Alcohol Spectrum Disorder (FASD) remains a major public health problem. This research employs a well-established mouse model to answer FASDrelated questions that are impossible to address in clinical studies. Focusing on stage of exposure-dependent facial and brain abnormalities, the results of this work promise to expand our understanding of the temporal dependence, interrelatedness, and mechanisms underlying birth defects that result from maternal alcohol use.
Prenatal alcohol effects on brain development and how it relates to behavior
 Our work focuses on the effects of prenatal alcohol exposure on brain development. Different parts of the brain, and connections between them, have different functions (e.g., language, decision making, learning), and some
Our work focuses on the effects of prenatal alcohol exposure on brain development. Different parts of the brain, and connections between them, have different functions (e.g., language, decision making, learning), and some
functions are more affected in individuals with FASD than others. Our work also shows that the large changes that normally occur in the brain through young adulthood are altered in those with FASD. These changes in the brain
are known to be affected by experiences, experiences that may happen long after the brain is damaged by alcohol. Knowing which brain areas are most affected by prenatal alcohol will help us learn if brain development can be improved by altering experiences with specific treatments. While prevention is ideal, our findings suggest that
interventions may improve the lives of children with FASD and their families, even when given later in life.
Elizabeth Sowell
Mapping the Brain, the Face, and Neurocognitive Function in FASD (U01) U01AA017122-07 | Children’s Hospital Los Angeles
Moving forward with the new theme for our project “brain connectivity in FASDs,” we propose to build on our work investigating the impact of prenatal alcohol exposure (AE) on the long-term development of the brain. Specifically, we will leverage advances in the field to implement two new imaging modalities to complement our existing work to address how structural and functional brain connectivity is affected, both cross-sectionally and longitudinally and how connectivity relates to cognitive function and facial dysmorphology in children with AE. The ultimate goal of this renewal application remains the same, to determine if innovative techniques can be used to identify brain alterations, neurobehavioral deficits and facial characteristics and relationships between these variables to help further define/diagnose prenatal alcohol spectrum disorders (FASD). This timely project will apply a cutting-edge multimodal Magnetic Resonance Imaging (MRI) protocols and sophisticated image analyses to study 240 children (120 with AE)
longitudinally studied across 4 data collection sites. While this brain imaging project can independently achieve some of the goals of the CIFASD by identifying brain structural and functional connectivity abnormalities across the broad spectrum of FASDs, critically this funding opportunity will allow the assessment of relationships between the brain, neurocognitive deficits and facial dysmorphology leveraging our active collaborations with the neurobehavioral project (Mattson PI), the facial imaging project (Foroud PI), and the dysmorphology core (Jones PI). Controlled animal studies are essential to determine timing and dosages of prenatal alcohol that result in FASDs, but human imaging studies are essential to corroborate anatomical findings across species that will be facilitated by projects in the basic sciences (Sulik PI). Thus, by capitalizing on existing CIFASD infrastructure, this project will provide critical new knowledge concerning the effects of AE on brain structure, function, behavior assessed cross-sectionally; in
addition, longitudinal examinations will determine if AE effects are static, or changing during childhood and adolescence. New knowledge gained through investigations during the renewal period could have strong implications for potential future interventions, given that the brain changes dramatically in even typically developing kids, and we know from animal studies that environment and experience (enriched environment vs. deprived, etc.) shape the way the brain develops. If trajectories of developmental change throughout childhood are impacted by damage resulting from teratogenic exposure to alcohol in utero, it could be possible to ultimately shift trajectories for the better in these children through interventions targeted towards brain regions and cognitive functions most affected by AE.
PUBLIC HEALTH RELEVANCE: Understanding the neurobiological consequences of prenatal alcohol exposure is a fundamental first step for determining when and how to best intervene to improve cognitive and functional outcomes of affected individuals. While prenatal alcohol exposure is the leading preventable cause of intellectual disabilities, the prevalence of fetal alcohol syndrome is not declining, and the prevalence of those negatively affected by prenatal alcohol exposure, but without the defining facial dysmorphology to warrant the full FAS diagnosis is likely much higher. Thus, while prevention is key, developing intervention strategies is still a priority, and the brain imaging investigations proposed here can help drive these effort forward.
Prenatal alcohol effects on behavior and cognition
 We are working to identify the pattern of behavioral changes caused by prenatal alcohol exposure. We measure behavior and mental skills, such as memory, in children with FASD. Parents also provide information about their child’s behavior. We also test children with other developmental problems to find out which behavior changes are specific to FASD and which are shared with other conditions. We hope that this understanding can help us develop
We are working to identify the pattern of behavioral changes caused by prenatal alcohol exposure. We measure behavior and mental skills, such as memory, in children with FASD. Parents also provide information about their child’s behavior. We also test children with other developmental problems to find out which behavior changes are specific to FASD and which are shared with other conditions. We hope that this understanding can help us develop
a faster and better way for children with FASD to be identified by health care professionals. Finally, we are studying behavior in younger children so that we can identify affected children as early as possible. Recognizing the pattern of behavioral changes in FASD and identifying affected children early could enhance treatment and improve the lives of children with FASD.
Sarah Mattson
A Multisite Neurobehavioral Assessment Of Fetal Alcohol Spectrum Disorders U01AA014834-10 | San Diego State University
The proposed study is relevant to the overall goal of the CIFASD funding opportunity of “further refining definitive diagnoses of FASD at different stages of the lifespan based on biological, physical, neurological, or behavioral assessment, or a combination thereof.” It is also directly in line with three of the specific research areas identified in the RFA: (1) Enhanced understanding of the neurobehavioral phenotype of FAS and ARND, (2) Diagnosis of FASD, and (3) Early case identification. Although nearly 40 years have passed since the initial clinical delineation of fetal alcohol syndrome (FAS), understanding of the full spectrum of effects of prenatal alcohol exposure (AE) is incomplete. Even though many children with AE experience significant neurobehavioral and adaptive difficulties that are related to their exposure history, for a majority of the cases, the link between those difficulties and AE may go unrecognized. Given that early identification has been linked to improved prognosis, it is critical to improve identification of alcohol-affected children with a sensitive, clinically utilizable neurobehavioral profile. Recent research, including that funded by CIFASD, has focused on the specificity of alcohol’s effects by comparing children with AE and those with attention-deficit/hyperactivity disorder (ADHD). While this research has documented important similarities and differences between AE and ADHD, comparisons with a more heterogeneous contrast group may increase the generalizability of the profile as well as detect deficits that are common between neurodevelopmental conditions and those that are unique to the effects of prenatal alcohol exposure. Finally, previous work on the neurobehavioral profile has focused primarily on the executive function domain, however, preliminary data and previous research suggests that including measures of memory function may strengthen the existing profile. The overarching aim of the current proposal is to develop and implement clinically relevant and feasible measurement tools to accurately identify children who are affected by AE. The proposed study will build on our existing database derived from data collected from children between 8-16 years of age, across multiple clinical sites and expand it to include two age groups (5-7 years and 10-16 years) to improve early identification, as well as assess an additional neuropsychological domain (memory). Data will be collected from children with AE, non-exposed controls and heterogeneous clinical contrast subjects at both new and existing sites. This research will improve understanding of fetal alcohol spectrum disorders (FASD) by continuing ongoing efforts to develop and refine a sensitive and specific neurobehavioral profile. These data will be combined with data from other CIFASD projects to develop models that accurately capture the greatest number of affected children using multidisciplinary methodology.
PUBLIC HEALTH RELEVANCE: This project is directly relevant to public health concerns surrounding the effects of heavy prenatal alcohol exposure, defining the profile of neurobehavioral effects that are specific to alcohol, and improving identification of alcohol-affected individuals at younger ages. Improved identification and delineation of these features, especially in younger children, will ultimately lead to improved treatment.
Early identification of FASD and nutrition
 Our work focuses on learning more about nutrition in both the mother and her child with an FASD and whether improving nutrition can reduce FASD. Our first goal is to prevent FASD by urging all women to avoid alcohol while planning and during the entire pregnancy. However, unplanned pregnancies continue to happen. Our work shows that better nutrition in the mother before and during pregnancy is good for the baby, and that prenatal vitamin and mineral supplements during pregnancy may improve the infant’s growth and learning in early life, even if they have been exposed to prenatal alcohol. We continue to explore how the diet of a child with an FASD might affect that child’s growth and development. This work will help us learn more about what interventions might be helpful for children to help them reach their potential throughout life.
Our work focuses on learning more about nutrition in both the mother and her child with an FASD and whether improving nutrition can reduce FASD. Our first goal is to prevent FASD by urging all women to avoid alcohol while planning and during the entire pregnancy. However, unplanned pregnancies continue to happen. Our work shows that better nutrition in the mother before and during pregnancy is good for the baby, and that prenatal vitamin and mineral supplements during pregnancy may improve the infant’s growth and learning in early life, even if they have been exposed to prenatal alcohol. We continue to explore how the diet of a child with an FASD might affect that child’s growth and development. This work will help us learn more about what interventions might be helpful for children to help them reach their potential throughout life.
Christina Chambers
Early Identification of Affected Children and Risk Factors for FASD in Ukraine U01AA014835-10 | University of California San Diego
It is estimated that Fetal Alcohol Spectrum Disorders (FASD) is the most common neurodevelopmental disability among children throughout the world. Furthering our understanding of the factors that potentially modify risk for FASD, continue to be critical gaps in
research that could drive prevention and intervention strategies throughout the world. A longitudinal cohort sample has been established in Ukraine during the current funding period with detailed documentation of prenatal alcohol exposure, maternal nutritional status, and FASD outcomes. In this same sample a multimicronutrient supplement intervention trial has been implemented. During the renewal period, we propose to examine the persistence of and/or the novel manifestation of beneficial neurobehavioral effects of the prenatal multimicronutrient intervention trial, to examine the role of postnatal child nutrition in FASD, an to explore genetic and epigenetic factors as biomarkers of maternal exposure and risk modification among alcohol consuming pregnant women.
PUBLIC HEALTH RELEVANCE: Fetal Alcohol Spectrum Disorders (FASDs) are among the most common developmental disabilities throughout the world. Identification of effective methods for prevention and early intervention for FASD is critical. In a longitudinal study in Ukraine, earlier identification and prevention of FASD is being evaluated in four areas: a) the contribution of pre and postnatal nutritional factors to risk for FASD, b) development of a preschool neurobehavioral testing battery appropriate for early identification of affected children c) the utility of a novel and objective biomarker of exposure to alcohol, and d) exploration of genetic and epigenetic factors that may modify risk for FASD in mother/child pairs.
CIFASD data management
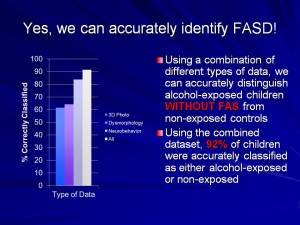
 The effects of prenatal alcohol exposure on children are complex. Alcohol affects physical, brain and behavioral development and each child may be affected in different ways. We bring together all the data from the studies of children with FASD and animal models so that we can develop an accurate picture of how alcohol during pregnancy affects the child. We are also examining how the diet of both mother and child affects FASD. This combined research will help us identify which children have been affected by alcohol, what effects were caused by alcohol, and what we can do to improve their lives. We want to make sure that we help identify the right solutions that will improve the life of your child.
The effects of prenatal alcohol exposure on children are complex. Alcohol affects physical, brain and behavioral development and each child may be affected in different ways. We bring together all the data from the studies of children with FASD and animal models so that we can develop an accurate picture of how alcohol during pregnancy affects the child. We are also examining how the diet of both mother and child affects FASD. This combined research will help us identify which children have been affected by alcohol, what effects were caused by alcohol, and what we can do to improve their lives. We want to make sure that we help identify the right solutions that will improve the life of your child.

William K. Barnett
Informatics Core, Collaborative Initiative on Fetal Alcohol Spectrum Disorders U24AA014818-10 | Indiana University Bloomington
The CIFASD Informatics Core provides a research data resource to be used by investigators to explore the relationships of face, brain, and behavior to improve the diagnosis of FAS and work towards therapies for alcohol exposed children. The Informatics Core has the following specific aims for the next five years: Aim 1: Continued cyber infrastructure support. The Informatics Core will consult and collaborate with CIFASD clinical projects, cores, and developmental projects to support the existing data submission to the CIFASD Central Repository, manage those data, and make those data available for use to accomplish CIFASD goals. Aim 2: Collection of additional data sets. The Informatics Core will securely bring new data sets into the CIFASD Central Repository for cross-study and cross-modality data analysis amongst CIFASD projects. Aim 3: Support of affiliated projects. The Informatics Core will develop technical solutions that will allow the comparison of data between CIFASD and affiliated projects. The Informatics core will use the following methods to support the goals of this project and CIFASD: The ongoing management of standardized data in the CIFASD Central Repository will provide core consortium resources including; data input and bulk upload tools, a data dictionary to standardize terms across studies, a cross query tool so that data from multiple studies can be integrated for synthetic studies, and data browsing tools that enable investigators to ensure data quality. Expert consultation will support the input, export, and integration of data in the Central Repository to lower the barrier of use for investigators and improve data quality of synthetic studies. This consultation will also regularly report on data in the Central Repository to track consortium progress goals. Expert software engineering will provide new input tools and modify existing tools to support the changing needs of CIFASD projects as well as new research projects that are added as part of the third Phase of the CIFASD program.
PUBLIC HEALTH RELEVANCE: The goal of the CIFASD Informatics Core is to support research on the relationships efface, brain, and behavior in order to improve the diagnosis of Fetal Alcohol Spectrum Disorders and work towards therapies for alcohol exposed children. By providing a central, standardized data resource for CIFASD, the Informatics Core will provide a means by which comprehensive diagnostics and therapies can be developed.
Genes and FASD
 Understanding the genes that increase risk to FASD can help improve diagnosis and treatment of affected children. Our research studies genes in the zebrafish, because the genes are similar to those of humans. With the zebrafish, we can identify genes that may increase risk to FASD and study their complex interactions much faster than we can in humans. Our studies will also help us identify genes that protect against the harmful
Understanding the genes that increase risk to FASD can help improve diagnosis and treatment of affected children. Our research studies genes in the zebrafish, because the genes are similar to those of humans. With the zebrafish, we can identify genes that may increase risk to FASD and study their complex interactions much faster than we can in humans. Our studies will also help us identify genes that protect against the harmful
effects of alcohol during development. Once we have identified these genes, we will study the function of the genes to understand how they affect the actions of alcohol on the fetus. We will work together with other CIFASD researchers to better understand the role of genes in FASD.
Johann Eberhart
Genetic approaches to understand variability in FASD facial and neural phenotypes | The University of Texas at Austin
In our developmental project we seek to identify gene-ethanol interactions using the zebrafish as a genetically amenable model organism. We will use two genetic approaches to identify and characterize gene-ethanol interactions that predispose embryos to ethanol-induced defects. In Specific Aim 1 we will characterize mutants from a forward genetic screen for ethanol-sensitive loci regulating facial/neural development. We have isolated 9 ethanol sensitive zebrafish mutants from a forward genetic screen. In this aim, we will determine the molecular nature of the genetic lesion and characterize the cause of the facial phenotype. Because facial and neural defects are often coincident in FASD we will also use marker and transgenic analyses to screen for neural defects. In Specific Aim 2 we will screen community resources to identify ethanolsensitive loci involved in facial/neural development. The Zebrafish International Resource Center (ZIRC) houses and distributes zebrafish genetic resources. We have identified 27 facial and/or neural mutant strains housed in ZIRC that are available alive and have not been screened for ethanol sensitivity. Here, we screen these mutants to determine if ethanol exacerbates the mutant phenotype or causes haploinsufficiency in eterozygotes. Because the mutation in these lines is understood, we will quickly be able to analyze the mechanism of these individual gene-ethanol interactions. The gene regulatory networks driving facial and neural development are highly conserved between mammals and zebrafish. Therefore,the geneethanol interactions that we identify in zebrafish are likely to modulate FASD phenotypes in human. Understanding the nature of these interactions will help in genetic counseling as well as diagnosis and designing possible treatments for FASD.
FASD, stress and daily rhythms
 Prenatal alcohol exposure can disrupt sleep, stress responses and mood (depression). Our research focuses on how alcohol disrupts stress and daily rhythms, immune responses, and risk for cancer. This research has
Prenatal alcohol exposure can disrupt sleep, stress responses and mood (depression). Our research focuses on how alcohol disrupts stress and daily rhythms, immune responses, and risk for cancer. This research has
shown that although the body can recover from damage or disease, the loss of some nerve cells may permanently impair stress and immune systems throughout life. In addition, prenatal alcohol may alter genes important in
these systems and we are measuring these genes in blood samples from children with and without FASD. These gene changes may serve as markers that help us identify children with FASD as early as possible.
Dipak K. Sarkark
Circadian Genes, Stress Axis and Fetal Alcohol Spectrum Disorder | Rutgers University
Recently, it has been shown that disruption of normal circadian rhythmicity is associated with various pathologies including sleep disorders, stress abnormality and depression. Because clinical sleep-wake disturbances, stress abnormality and depression have been observed in human neonates, children, and adolescents following prenatal exposure to alcohol, Dipak Sarkar proposes to measure Per genes (Per1 and Per2) methylation and gene expression as well as plasma cortisol levels using the DNA and plasma samples of longitudinal studies with FASD and non-FASD patient children from the Collaborative Initiative on Fetal Alcohol Spectrum Disorders. The questions to be addressed in his proposal are: Do methylation and expression levels of Per genes correlate with the stress abnormality (high cortisol)? Can these methylation sites and patterns be used as biomarkers that predict future endophenotypes of stress abnormalities (e.g., anxiety, alcohol binging)? Furthermore, if Per genes hypermethylation is involved in stress abnormalities, whether this is manifested in patients with FASD. These studies will be crucial in determining the stability of an identified epigenetic signature of FASD patients and whether epigenetic signatures identified allow for earlier identification of alcohol-related birth outcomes.
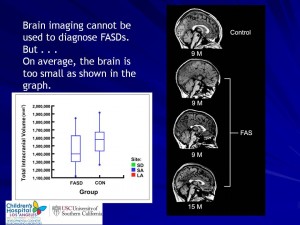
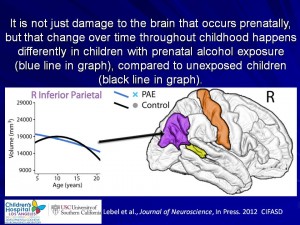
Prenatal alcohol effects on early brain development
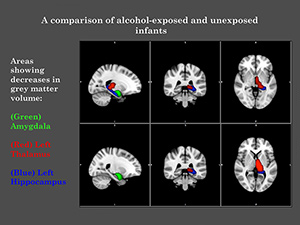
 This research project aims to show how sophisticated brain-imaging techniques (and 3D-facial imaging) may be able to identify early FASD-related abnormalities with the brain, before developmental and behavioural problems are observed. This research project also looks at the long-term effects of alcohol-exposure on a young child’s developing brain – birth to 2-years old.
This research project aims to show how sophisticated brain-imaging techniques (and 3D-facial imaging) may be able to identify early FASD-related abnormalities with the brain, before developmental and behavioural problems are observed. This research project also looks at the long-term effects of alcohol-exposure on a young child’s developing brain – birth to 2-years old.
Drinking alcohol during pregnancy can result in harmful consequences for the unborn child. This ‘in utero‘ exposure to alcohol can physically alter regions of the child’s brain, which can already be observed in newborns. The brain is developing rapidly at this time, with significant changes occurring in its structural and functional networks. Making this both a vulnerable and very important time, in the development of the young brain.
This project is a step towards forming an effective strategy to identify and detect early signs for a possible FASD diagnosis, by making use of sophisticated brain-imaging techniques to detect early abnormalities with the brain. An early diagnosis will allow for early interventions and treatment plans to be setup. Early treatment may lead to better long-term outcomes for these vulnerable children.
Kirsty Donald
A comparison of alcohol-exposed and unexposed infants | University of Cape Town
Prenatal alcohol effects on immune function
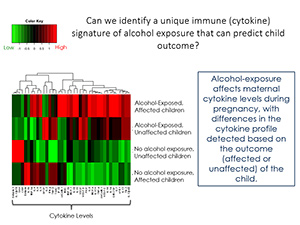
 The immune system plays a critical role in a person’s overall health, and there is growing evidence for immune system involvement in healthy brain function. This begins during fetal life, when immune system factors, known as cytokines, play a key role in shaping brain development. Prenatal exposure to alcohol can adversely impact fetal immune system development, and in turn, fetal brain development, with implications not only for long-term health outcomes, but also for behavior, cognition, and adaptive functioning. In our study, we are measuring cytokine levels in alcohol-consuming and abstinent pregnant women and their children, with the aim of identifying an immune signature that is unique to alcohol exposure, and that may be linked to maternal immune function. Our findings will have implications for understanding long-term health outcomes in children with Fetal Alcohol Spectrum Disorder (FASD), and may point the way toward the development of novel interventions for these children.
The immune system plays a critical role in a person’s overall health, and there is growing evidence for immune system involvement in healthy brain function. This begins during fetal life, when immune system factors, known as cytokines, play a key role in shaping brain development. Prenatal exposure to alcohol can adversely impact fetal immune system development, and in turn, fetal brain development, with implications not only for long-term health outcomes, but also for behavior, cognition, and adaptive functioning. In our study, we are measuring cytokine levels in alcohol-consuming and abstinent pregnant women and their children, with the aim of identifying an immune signature that is unique to alcohol exposure, and that may be linked to maternal immune function. Our findings will have implications for understanding long-term health outcomes in children with Fetal Alcohol Spectrum Disorder (FASD), and may point the way toward the development of novel interventions for these children.
Joanne Weinberg
Identifying an immune signature characteristic of FASD: Implications for possible immune-based intervention strategies | The University of British Columbia
Abnormal immune system function during development can lead to aberrant brain development and immune dysregulation, and, in turn, increased vulnerability to diseases or disorders later in life, including infections, cardiovascular disease, diabetes, cognitive and behavioral problems and mental health disorders. Importantly, prenatal exposure to alcohol adversely affects development of the fetal immune system, and in turn, many of these diseases/disorders are highly prevalent in individuals with Fetal Alcohol Spectrum Disorder (FASD). Yet despite identification of immune deficits associated with prenatal alcohol exposure, clinical and pre-clinical immune-related research remains an understudied area in the FASD field. The present study is motivated by our ontogenetic profiling of the immune system in our rat model of prenatal alcohol exposure, which found increased circulating and brain levels of proinflammatory cytokines from birth to weaning age, suggesting that some of the negative effects of prenatal alcohol exposure may originate from this early proinflammatory bias. In collaboration with Dr. Christina Chambers and her team, our project will use samples from the longitudinal study in Ukraine to perform a comprehensive assessment of the immune system in cohorts of pregnant women who did or did not consume alcohol during pregnancy their children; specifically, we will measure plasma levels of 40 pro- and anti-inflammatory cytokines. Identification of unique immune signatures in both pregnant women and their children will provide valuable information regarding how the immune system may mediate at least some of the adverse effects of in utero alcohol exposure. Furthermore, we aim to identify cytokine signatures in the mother that could used to predict child outcome (affected vs. unaffected by alcohol). Moreover, cytokine profiles could be used as a biomarker of prenatal alcohol exposure, as well as in design of targeted immune-based interventions for FASD.
PUBLIC HEALTH RELEVANCE: Enduring alterations in immune system function are associated with a wide range of physical, cognitive, behavioral, and mental health problems in adulthood. Identification of specific immune system alterations associated with prenatal alcohol exposure could serve as a biomarker and is a critical step towards establishing more targeted interventions and/or treatments for affected individuals.