Research Projects and Components
- Administrative Resource of the CIFASD (Edward Riley, PI)
- FASD Diagnostic Telemedicine Resource (Miguel del Campo, PI)
- CIFASD Data Coordination Resource (Leah Wetherill, PI)
- Cardiovascular Disease in Fetal Alcohol Spectrum Disorder (Caroline Burns and Geoff Burns, MPIs)
- Whole body Effects of PAE across the Life Span: Early Markers of & Clinical Interventions for Children and Adolescents (Christina Chambers, PI)
- A Multisite Study of Prenatal Alcohol Exposure: Inflammation and Endocrine Dysfunction in Adulthood (Claire Coles PI)
- Designing a Hybrid Intervention Strategy to Reduce Alcohol Exposed Pregnancies (Ralph DiClemente, PI)
- Assessment of Fetal Alcohol Spectrum Disorders (FASD) Using Novel Web-Based Tools (Sarah Mattson, PI)
- Leveraging Technology to Increase Quality of Life for FASD across the Lifespan (Christie L. McGee Petrenko and Cristiano Tapparello, MPIs)
- Defining Translational Approaches for the Image-Based Detection of Prenatal Alcohol Exposure (Michael Suttie, PI)
- tDCS and Cognitive Training as a Neurodevelopmental Intervention in FASD (Jeff Wozniak, PI)
- Lifelong Impact of PAE on Stem Cell Dynamics and Cellular Aging (Amanda Mahnke, PI)
- Biomarkers in Children with FASD that Predict Neurobehavioral Performance (Annika Montag and Christine Austin, MPIs)
- Mobile Health Tools to Promote Health in Adults With FASD (Christie L. McGee Petrenko and Cristiano Tapparello, MPIs)
- Choline Polymorphisms in FASD (Susan M. Smith, PI)
Administrative Resource of the CIFASD
PI: Edward Riley
Administrative Resource of the CIFASD U24 AA014811 | San Diego State University
 This project brings experts together to better understand how prenatal alcohol affects the developing baby, so that we can better treat and prevent fetal alcohol spectrum disorders (FASD). Our goals are to better identify individuals who have been exposed to alcohol prenatally, to determine factors that increase or reduce risk to FASD, to understand the effects of prenatal alcohol exposure across the lifespan, and to develop treatments for FASD. We are investigating new mobile eHealth approaches to better achieve these goals, providing scalable tools that can be used to help identify and treat individuals with FASD, even in remote locations.
This project brings experts together to better understand how prenatal alcohol affects the developing baby, so that we can better treat and prevent fetal alcohol spectrum disorders (FASD). Our goals are to better identify individuals who have been exposed to alcohol prenatally, to determine factors that increase or reduce risk to FASD, to understand the effects of prenatal alcohol exposure across the lifespan, and to develop treatments for FASD. We are investigating new mobile eHealth approaches to better achieve these goals, providing scalable tools that can be used to help identify and treat individuals with FASD, even in remote locations.
The Collaborative Initiative on Fetal Alcohol Spectrum Disorders (CIFASD) is a multidisciplinary, international consortium of research projects and resources charged with improving prevention, diagnosis, and treatment of FASD. The Administrative Core/Resource (AdminC) of CIFASD has led this consortium since it was first established in 2003 and plays a critical leadership role in coordinating research efforts and ensuring progress toward consortium goals. CIFASD consists of multiple collaborative components including three resource U24s (AdminC, Diagnostic-Telemedicine, and Data Coordination), eight clinical and/or translational U01 projects, and two exploratory UH2 projects. One goal of CIFASD is to improve identification of individuals affected by prenatal alcohol exposure (PAE). This will be accomplished by assessing biomarkers of exposure or effects, such as immune, HPA, and miRNA profiles. In addition, CIFASD is implementing novel tools, such as 3D facial and neurobehavioral screening to improve identification of FASD remotely through eHealth, providing more universal accessibility. A focus on telemedicine provides access to dysmorphology evaluations in more diverse and remote populations. CIFASD continues to study the effects of PAE on the whole body across the lifespan. Previous results from CIFASD suggest that PAE may be a risk factor for numerous health issues (e.g., hypertension and diabetes). By using interrelated clinical and preclinical research approaches, health outcomes and markers will be examined at several ages. CIFASD continues work on interventions to mitigate the damaging effects of PAE including a trial on tDCS, and expansions of eHealth initiatives to provide interventions to assist affected individuals and their families. To improve prevention of maternal alcohol consumption and reduce risk for FASD, a hybrid prevention strategy will be investigated. Implementation is a new focus of CIFASD; as unless tools created in CIFASD are utilized clinically, their potential won’t be fully realized. Collectively, CIFASD projects will move the FASD field forward, improving prevention, diagnosis, and treatment and with the technology available in eHealth serve a wide population. The Specific Aims of the AdminC are: 1) Provide scientific and administrative direction, leadership, and oversight; 2) Facilitate communication among the projects and result dissemination; 3) Assist with data management; 4) Provide annual progress evaluations, and 5) Support projects by providing outreach, eHealth, and implementation science resources and services. The AdminC supports the interactive framework of CIFASD by regular communications with all members through monthly and biannual meetings; leading the Steering Committee in the establishment and implementation of CIFASD priorities and policies; supporting the Science Advisory Board’s evaluations of progress on each component and the overall mission of CIFASD; serving as the liaison between the consortium’s project scientists and its NIAAA advisors; and supporting outreach, eHealth, and implementation efforts.
The consequences resulting from prenatal alcohol exposure are wide-ranging and a major and costly public health problem. The goal of the CIFASD consortium is to enhance the identification and treatment of FASD utilizing a multidisciplinary approach consisting of both basic science and clinical research. The collaborations among research projects will allow CIFASD to improve the diagnosis and treatment of FASD, moving the field forward in ways that could not be easily achieved by individual projects.
FASD Diagnostic Telemedicine Resource
PI: Miguel del Campo
FASD Diagnostic Telemedicine Resource U24 AA014815 | University of California, San Diego
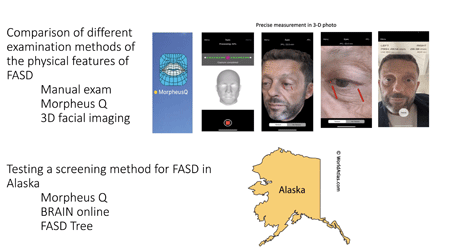
 Effective screening and diagnoses of fetal alcohol spectrum disorders (FASD) is challenging, limiting our ability to target prevention and intervention strategies. This core resource first ensures that the physical components of FASD are identified accurately and consistently in all CIFASD projects involving human subjects. With the goal of improving precision and increasing access to screening and diagnosis, we will use standardized procedures to train all examiners involved in these projects and will closely supervise their performance. We will also determine of new automated tools can accurately identify physical features associated with prenatal alcohol exposure, in an effort to increase accessibility and accuracy of diagnoses. Finally, we will test a novel system for screening of FASD in Alaska, with the goal of providing a screening system for remote underserved areas.
Effective screening and diagnoses of fetal alcohol spectrum disorders (FASD) is challenging, limiting our ability to target prevention and intervention strategies. This core resource first ensures that the physical components of FASD are identified accurately and consistently in all CIFASD projects involving human subjects. With the goal of improving precision and increasing access to screening and diagnosis, we will use standardized procedures to train all examiners involved in these projects and will closely supervise their performance. We will also determine of new automated tools can accurately identify physical features associated with prenatal alcohol exposure, in an effort to increase accessibility and accuracy of diagnoses. Finally, we will test a novel system for screening of FASD in Alaska, with the goal of providing a screening system for remote underserved areas.
Prenatal alcohol exposure is estimated to impact 1-5% of children in the US alone. Yet, many individuals who have been exposed prenatally to alcohol and suffer from fetal alcohol spectrum disorders (FASD) fail to be recognized. This failure is due, in large part, to a paucity of specialized clinics and expert dysmorphologists who are trained to identify FASD. Access to appropriate diagnostic expertise is particularly limited in remote areas. Unfortunately, if individuals with FASD are not recognized and diagnosed, they do not receive critically needed services nor can they access potential interventions early in life, when intervention is most likely to be effective. The CIFASD5 Diagnostic-Telemedicine Resource (DTR) will ensure consistent and accurate assessment of the physical characteristics of FASD across the CIFASD research sites. In addition, the DTR will address the critical need for increased diagnostic capacity through training of non-expert practitioners across CIFASD5 sites using telemedicine-based methods. In the previous CIFASD4 iteration, telemedicine approaches were tested for this purpose in a small sample of clinicians and found to be an effective method for training and monitoring of new examiners. In CIFASD5, this method will be extended to multiple sites and be employed in the evaluation of over 1,800 children and adults. However, telemedicine alone is insufficient to expand capacity and ensure consistency and accuracy of diagnosis. To that end, several novel eHealth tools have been developed to assist in the detection of physical features associated with prenatal alcohol exposure. These tools hold promise in providing simple and efficient ways to screen and identify FASD. These include MorpheusQ, a smart-phone based app that automates facial feature detection; Face-to-Gene, a 2D facial image diagnostic aid used by clinical geneticists to screen for potential syndromes; and 3D facial image signatures. These tools are scalable and have the potential to improve screening and diagnosis across the globe, even in remote areas, such as in Alaska. However, the diagnostic accuracy of these tools needs to be systematically compared to standardized dysmorphological exams. It is essential to determine whether eHealth tools can effectively replace and/or improve traditional exams before they are widely implemented.
The primary goal of the CIFASD Consortium Diagnostic-Telemedicine Resource is to ensure consistent and accurate diagnoses of children and adults with fetal alcohol spectrum disorders (FASD) at study sites throughout the world. Because there is a critical shortage of health care practitioners who have the expertise to make this diagnosis, the Diagnostic-Telemedicine Resource will train new practitioners using telemedicine, aided by novel eHealth tools to perform these examinations. Finally, we will test these methods in delivering diagnostic services for FASD to remote areas of Alaska.
CIFASD Data Coordination Resource
PI: Leah Wetherill
CIFASD Data Coordination Resource U24 AA030169 | Indiana University – Purdue University at Indianapolis
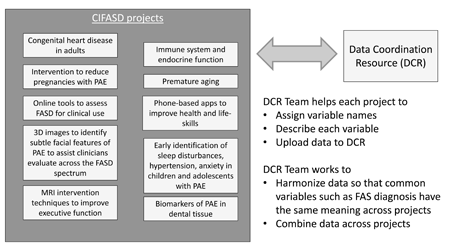
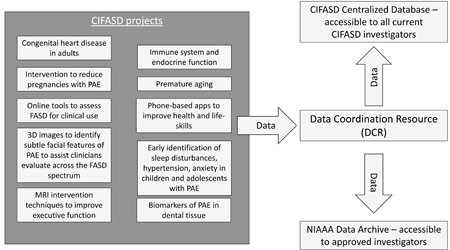
 The most significant contributions to understanding health-related issues have resulted from scientists sharing their data and working together. Fully understanding the impact of prenatal alcohol exposure (PAE), requires the same type of collaboration. CIFASD investigators study how PAE affects the brain, behavior, immune system, physical health, and physical features. They are also trying to identify markers that could indicate conditions associated with PAE, and to find ways to help individuals and families cope with the effects from PAE. Each investigator uploads their data to the Data Coordination Resource, where we harmonize and store it in a safe, central online location for CIFASD investigators. We also transfer the data to the National Institute on Alcohol Abuse and Alcoholism Data Archive, where approved researchers can explore relationships in this wealth of data. Our team is dedicated to providing safe and easy access to this invaluable data, to better understand the effects of PAE throughout a person’s life.
The most significant contributions to understanding health-related issues have resulted from scientists sharing their data and working together. Fully understanding the impact of prenatal alcohol exposure (PAE), requires the same type of collaboration. CIFASD investigators study how PAE affects the brain, behavior, immune system, physical health, and physical features. They are also trying to identify markers that could indicate conditions associated with PAE, and to find ways to help individuals and families cope with the effects from PAE. Each investigator uploads their data to the Data Coordination Resource, where we harmonize and store it in a safe, central online location for CIFASD investigators. We also transfer the data to the National Institute on Alcohol Abuse and Alcoholism Data Archive, where approved researchers can explore relationships in this wealth of data. Our team is dedicated to providing safe and easy access to this invaluable data, to better understand the effects of PAE throughout a person’s life.
Almost 10% of pregnancies world-wide were reported to have prenatal alcohol exposure (PAE) (Popova, Lange, Probst, Gmel, & Rehm, 2017). In the United States a conservative estimate of Fetal Alcohol Spectrum Disorders (FASD) in first-grade children was 1-5% (May et al., 2018). These numbers are expected to increase, given the slight but steady rise in alcohol use among pregnant women (Denny, Acero, Terplan, & Kim, 2020). NIAAA established the Collaborative Initiative on the Fetal Alcohol Spectrum Disorders (CIFASD) to inform and develop effective interventions and treatment approaches for FASD through a highly integrated, multidisciplinary research program involving basic scientists and clinical investigators. Central to the success of CIFASD is strong central coordination provided through the Administrative Resource and a Data Coordination Resource (DCR). As part of CIFASD5, the DCR will collaborate with all projects and resources within the consortium to provide support and management with respect to data collection, storage and sharing. Based on this central role, the DCR will coordinate the efforts of the CIFASD5 investigators conducting human subjects research to meet the requirement of depositing data into the NIAAA Data Archive (NDA) and will serve as the primary contact with NDA in the biannual transfer of data to the NDA. The DCR will be responsible for ensuring data sharing in CIFASD5 conforms with NIH Policy. In addition, the DCR will support the efforts of the Administrative Resource in tracking progress of CIFASD5. To meet these goals, the DCR proposes the following specific aims: 1. Create a Data Coordination Resource (DCR) database for CIFASD5. 2. Serve as the CIFASD5 point of contact for the NIAAA Data Archive. 3. Expand the functionality of the DCR database. 4. Support the distribution of archived CIFASD data. 5. Maintain an online registry of consented CIFASD research participants available for recruitment into other projects.
The Data Coordination Resource will create an integrated database consisting of a vast array of data collected from the CIFASD projects in an effort to accurately recognize those with prenatal alcohol exposure (PAE), improve interventions to mitigate the effects of PAE, better understand the effects of PAE across the lifespan, and to reduce the incidence of PAE. These data will be shared with the broader research community, contributing to further understanding the effects of PAE and enhance future clinical and translational research.
Cardiovascular Disease in Fetal Alcohol Spectrum Disorder
MPIs: Caroline Burns and Geoff Burns
Cardiovascular Disease in Fetal Alcohol Spectrum Disorder U01 AA030185 | Boston Children’s Hospital
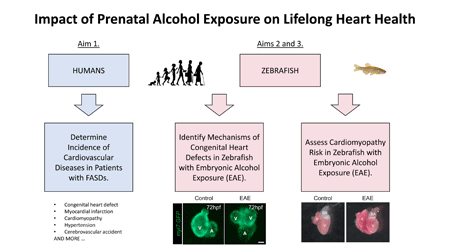
 Early stages of heart formation occur within the first weeks of fetal development, making the heart vulnerable to damage caused by alcohol exposure early in pregnancy. Many patients with FASD are born with visible heart problems called congenital heart defects. However, those born without visible heart problems may still have hidden issues with their hearts that will become a problem later in life. Importantly, there is a poor understanding of cardiovascular health outcomes in adult patients with FASD. We will investigate the incidence of cardiovascular diseases in patients with FASD across the life span. Results from these studies will help us know whether being exposed to alcohol before birth is a risk factor for adult heart problems and whether early intervention could help. We will also use a zebrafish model of prenatal alcohol exposure to determine the impacts of alcohol exposure on heart structure, function, and gene expression.
Early stages of heart formation occur within the first weeks of fetal development, making the heart vulnerable to damage caused by alcohol exposure early in pregnancy. Many patients with FASD are born with visible heart problems called congenital heart defects. However, those born without visible heart problems may still have hidden issues with their hearts that will become a problem later in life. Importantly, there is a poor understanding of cardiovascular health outcomes in adult patients with FASD. We will investigate the incidence of cardiovascular diseases in patients with FASD across the life span. Results from these studies will help us know whether being exposed to alcohol before birth is a risk factor for adult heart problems and whether early intervention could help. We will also use a zebrafish model of prenatal alcohol exposure to determine the impacts of alcohol exposure on heart structure, function, and gene expression.
The incidence of congenital heart defects (CHDs) and cardiovascular disease (CVD) in patients with fetal alcohol spectrum disorder (FASD) are poorly characterized. Cardiovascular abnormalities may be common in FASD; however, comprehensive retrospective studies on lifetime CVD risk in adult patient cohorts have yet to be performed. Cellular and molecular mechanisms underlying FASD-mediated CHD and CVD are also largely unknown along with any biomarkers that would allow the patient population to be stratified based on CVD risk. Here we present preliminary data from our retrospective clinic cohort that demonstrate that females with FASD have an overall increase in CHD, myocardial infarction (MI) rate, and the likelihood of being diagnosed with any CVD in adulthood. Females with FASD also have significantly reduced ejection fraction relative to matched controls. These data suggest that FASD is a risk factor for CHD in newborns and CVD in adults. In a zebrafish model of embryonic alcohol exposure (EAE), we confirmed a primary defect in cardiomyocyte migration that causes subsequent functional and structural heart abnormalities, including contractility deficits and ventricular wall abnormalities that persist through adulthood. Our findings indicate that EAE zebrafish can serve as a model for lifelong cardiac function in the presence and absence of CHD. We propose three Specific Aims to test the central hypothesis that FASD patients have an increased incidence of CVD and that the zebrafish EAE model will uncover novel molecular mediators and biomarkers that explain and predict CVD risk. In Specific Aim 1, we will perform a retrospective study to determine CVD incidence in an adult FASD patient cohort, including CHD, hypertension, cardiomyopathy, MI, cerebrovascular accident, and embolism, as well as their association with other metabolic and inflammatory conditions. In Specific Aim 2, we will define molecular mechanisms underlying embryonic heart defects in a zebrafish EAE model by identifying and functionally evaluating the impact of molecular alterations in migratory myl7+ cardiomyocytes that form the cardiac cone through hypothesis-driven (PDGF pathway) and unbiased (bulk RNA-sequencing on FACS-isolated myl7+ cardiomyocytes) approaches. In Specific Aim 3, we will test the hypothesis that EAE adults with a CHD are susceptible to cardiac dysfunction and cardiomyopathy due to lasting alterations in cardiac structure, function, and molecular signature. Taken together, the proposed studies will provide fundamental insights into the cardiovascular health outcomes of patients with FASD, reveal novel molecular mediators of EtOH-induced CHDs, and identify biomarkers of adult cardiac dysfunction in EAE adults. Cardiovascular diseases may contribute significantly to morbidity and mortality in affected patients. By identifying which CVD outcomes impact FASD patients and what additional metabolic and inflammatory factors indicate risk, we will provide an opportunity for early intervention. Further, identification of molecular mediators of CHD and cardiomyopathy in a zebrafish model of EAE will allow us to expand our mechanistic understanding of the effects of PAE across the lifespan.
The long-term goal of this research is to decipher cardiovascular health outcomes in adults with FASD and to identify novel molecular mechanisms and biomarkers for ethanol-induced congenital heart defects and subsequent cardiac dysfunction in adulthood. We propose to accomplish these goals through a retrospective study on adult patients with and without FASD and by evaluation of a zebrafish model of embryonic alcohol exposure. Our findings will enable early identification or intervention in at-risk patients with FASD and expand our mechanistic understanding of the lasting effects of prenatal alcohol exposure on the heart.
Whole body Effects of PAE across the Life Span: Early Markers of & Clinical Interventions for Children and Adolescents
PI: Christina Chambers
Whole body Effects of PAE across the Life Span: Early Markers of & Clinical Interventions for Children and Adolescents U01 AA014835 | University of California, San Diego
 Emerging data suggest that adults with fetal alcohol spectrum disorders (FASD) have a wide range of co-occurring health challenges, including cardiovascular, metabolic, and mental health issues that may affect quality and length of life. This project will evaluate about 300 children and adolescents who have previously been followed for prenatal alcohol exposure in the U.S. The study will look for early clinical signs and biomarkers for these co-occurring health conditions. This work is essential to better understand how often and how early in the child’s life these health conditions are likely to occur. Findings can lead to recommendations for clinical assessments, as well as early interventions in children or adolescents with FASD to help prevent progression of disease. These results may improve overall health across the lifespan.
Emerging data suggest that adults with fetal alcohol spectrum disorders (FASD) have a wide range of co-occurring health challenges, including cardiovascular, metabolic, and mental health issues that may affect quality and length of life. This project will evaluate about 300 children and adolescents who have previously been followed for prenatal alcohol exposure in the U.S. The study will look for early clinical signs and biomarkers for these co-occurring health conditions. This work is essential to better understand how often and how early in the child’s life these health conditions are likely to occur. Findings can lead to recommendations for clinical assessments, as well as early interventions in children or adolescents with FASD to help prevent progression of disease. These results may improve overall health across the lifespan.
Numerous comorbidities have been identified as relatively common among children and adolescents with prenatal alcohol exposure (PAE) or fetal alcohol spectrum disorders (FASDs) including sleep disturbances, hypertension, abnormal eating behaviors, altered growth patterns, and comorbid depression and anxiety. In addition, emerging data suggest that several adult diseases of developmental origin, such as diabetes, atherosclerosis, cardiovascular and autoimmune disorders are over-represented and have earlier age at onset in adults with FASDs. It is imperative to better understand the prevalence and co-occurrence of these disorders as early as possible in the life course of children and adolescents with PAE, ideally at the sub-clinical stage, in order to intervene in clinically meaningful ways. Furthermore, in keeping with one of the primary goals of CIFASD5, findings of this study will directly inform future intervention targets in children and adolescents to help remediate, ameliorate or prevent progression of pre-clinical or clinical conditions identified in the study evaluations. We will also work collaboratively with other investigators in the CIFASD Consortium to interactively address the overall goals of the Consortium in improving diagnosis and treatment of FASD.
A better understanding of the prevalence and co-occurrence of relatively common comorbidities among children and adolescents with prenatal alcohol exposure (PAE) is essential in order to intervene in clinically meaningful ways. These complex comorbid conditions are often the most challenging, disruptive, and costly health issues that families with a child affected by alcohol must cope. This study will document the prevalence and co- occurrence of early-stage indicators of possible or probable future chronic disease in children/adolescents with PAE.
A Multisite Study of Prenatal Alcohol Exposure: Effects of Inflammation and Endocrine Dysfunction in Adulthood
PI: Claire Coles
A Multisite Study of Prenatal Alcohol Exposure: Effects of Inflammation and Endocrine Dysfunction in Adulthood U01 AA026108 | Emory University
 A lot is known about the effects of prenatal alcohol exposure (PAE) in children, but little is known about the long-term effects of PAE as individuals grow into adulthood. Our previous studies found that PAE negatively affects both physical and mental health in midlife, with many health problems occurring earlier than compared to the general population. Working at three North American sites, we are extending this research to examine how PAE impacts not only health, but also cognitive and adaptive function, as people age. We are also investigating how early life adversity, and lack of social resources and coping skills, may modify or increase the effects of PAE. Finally, we are investigating whether changes in key biological systems known to be affected by PAE may play a role in the increased risk for negative outcomes, pointing the way to possible novel intervention strategies to improve quality of life for this vulnerable population.
A lot is known about the effects of prenatal alcohol exposure (PAE) in children, but little is known about the long-term effects of PAE as individuals grow into adulthood. Our previous studies found that PAE negatively affects both physical and mental health in midlife, with many health problems occurring earlier than compared to the general population. Working at three North American sites, we are extending this research to examine how PAE impacts not only health, but also cognitive and adaptive function, as people age. We are also investigating how early life adversity, and lack of social resources and coping skills, may modify or increase the effects of PAE. Finally, we are investigating whether changes in key biological systems known to be affected by PAE may play a role in the increased risk for negative outcomes, pointing the way to possible novel intervention strategies to improve quality of life for this vulnerable population.
Although the impact of prenatal alcohol exposure (PAE) on early development has been well established, the Developmental Origins of Health and Disease (DOHaD) hypothesis suggests there may be longer- term consequences that increase the risk for adult diseases or disorders. Previously, it has been difficult to isolate the effects of PAE from those associated with other life experiences in affected individuals so that, often, the role of PAE may be overlooked. In fact, viewed from this context, PAE may be considered the first in a series of adverse exposures that result, eventually, in a higher risk for negative outcomes in this vulnerable population. Programing of fetal systems by PAE may alter the developmental trajectory and result in a sensitized organism that is vulnerable to later life challenges. The endocrine and immune systems have been found to be highly susceptible to programming by early life events, and together are thought to play key roles in linking early life adverse exposures with long-term health and functional outcomes. Thus, programming of these systems may be one mechanism by which PAE impacts adult health and neurobehavioral outcomes. In our ongoing CIFASD4 research negative impacts of PAE on physical and mental health outcomes in midlife were observed and attributable to both PAE and to associated early life adversity and social factors. Also noted were long-lasting, adverse changes in immune function, with inflammation starting in infancy and lasting into adulthood, suggesting an increased inflammatory burden over the life course. In the proposed study, we will test a “multiple hit” hypothesis that PAE results in a vulnerable organism that may be further impacted by social adversity and lack of resources/coping skills, resulting in immune and endocrine dysregulation that in turn, may be key drivers of early-onset health and neurobehavioral problems over the life course. 360 individuals representing a diverse sample of affected individuals from three sites will participate in a quasi-experimental study of PAE effects in midlife. In addition to measuring PAE/FASD and environmental conditions, we will use state-of-the-art methodologies to identify biomarkers (inflammatory, angiogenesis, vascular injury, metabolic, and neurodegenerative markers, transcriptional profiling of individual cells, and telomere length) of early-onset functional deficits including both physical and mental health. Specifically, within the context of the negative effects of social adversity and the positive influences of social resources and coping skills, we will evaluate: Aim 1. the role of immune and endocrine dysregulation in physical and mental health outcomes in adults with FASD/PAE; Aim 2 the impact of PAE and the possible role of PAE-induced immune and endocrine dysregulation on neurocognitive performance and markers of early-onset functional deficits in adults with FASD/PAE in comparison to age-matched controls and healthy older individuals.
In addition to physical, neurocognitive, physiological and behavioral abnormalities/deficits that occur following prenatal alcohol exposure (PAE), our findings in CIFASD 4 suggest negative impacts on both physical and mental health outcomes in midlife, with many health problems occurring earlier than would be expected in the general population. Findings also indicate that PAE is associated with long-lasting, adverse changes in immune and endocrine function, starting in infancy and lasting into adulthood. Using a “whole body, whole lifespan” approach, we will investigate how PAE-induced alterations in the immune and endocrine systems may be key drivers of early-onset health and functional deficits in adults with FASD.
Designing a Hybrid Intervention Strategy to Reduce Alcohol Exposed Pregnancies
PI: Ralph DiClemente
Designing a Hybrid Intervention Strategy to Reduce Alcohol Exposed Pregnancies U01 AA030187 | New York University
 This project is designed to reduce alcohol use among women in prenatal care as one strategy for preventing FASD. Women are recruited at clinics during routine prenatal care visits. Women who agree to participate and are identified as using alcohol complete a computerized assessment that measures alcohol use and other risk factors. Women also provide a small blood sample to assess alcohol use. After completing these assessments, women are assigned, by chance alone, to receive either the usual care from their obstetrician or the usual care from their Obstetrician plus a brief counseling intervention that includes nurse-delivered and computer-delivered motivational counseling. We will monitor alcohol use during prenatal care and assess birth outcomes at delivery. Women participating in the motivational counseling intervention are expected to reduce their alcohol use and have fewer poor birth outcomes.
This project is designed to reduce alcohol use among women in prenatal care as one strategy for preventing FASD. Women are recruited at clinics during routine prenatal care visits. Women who agree to participate and are identified as using alcohol complete a computerized assessment that measures alcohol use and other risk factors. Women also provide a small blood sample to assess alcohol use. After completing these assessments, women are assigned, by chance alone, to receive either the usual care from their obstetrician or the usual care from their Obstetrician plus a brief counseling intervention that includes nurse-delivered and computer-delivered motivational counseling. We will monitor alcohol use during prenatal care and assess birth outcomes at delivery. Women participating in the motivational counseling intervention are expected to reduce their alcohol use and have fewer poor birth outcomes.
Prenatal alcohol exposure (PAE) increases the risk for Fetal Alcohol Spectrum Disorders (FASD). To address this health threat, we propose an exciting collaboration between researchers at NYU, the University of Texas Health Science Center-Houston, Purdue University, and CIFASD to develop, implement, and evaluate an alcohol intervention for women in prenatal care. We propose a randomized controlled trial (N=600) to assess the efficacy of the alcohol intervention, relative to usual prenatal care only, in reducing (1) women’s alcohol use and 2) poor infant birth outcomes. We will recruit women at their first prenatal care visit. Eligibility criteria include (1) age >18 years; (2) ≤8 weeks gestation; (3) a positive rapid point-of-care ethyl glucuronide (EtG) urine test for recent alcohol use or self-reported alcohol use in the prior three weeks, and (4) written informed consent. Eligible women complete a baseline assessment by a Project Research Nurse (PRN) consisting of an ACASI and collection of blood spots for PEth analysis to assess alcohol consumption biologically. Subsequently, women are randomized to either (1) the intervention condition, the usual prenatal plus the alcohol intervention, or (2) the comparison condition, usual prenatal care only. Prenatal usual care involves clinicians assessing alcohol use and counseling women on alcohol-related risks. The alcohol intervention consists of (1) a self-paced computer-delivered component to enhance knowledge, norms, and motivation for alcohol reduction and (2) a PRN-delivered component to reinforce the computer-delivered content and address women’s questions. Both components are theory-driven, based on Motivational Enhancement Theory (MET), and use motivational strategies to promote alcohol reduction. The alcohol intervention is developed with guidance from multiple sources, including (1) the project’s Women’s Advisory Board, recruited from the proposed recruitment clinics who self-report alcohol use during pregnancy, and (2) project investigators and consultants with extensive experience and expertise in research and trial design, and developing and implementing SBIRTs for reducing alcohol use and other drugs among women, including pregnant women. The alcohol intervention is delivered three times: (1) following the baseline and the 2nd- and 3rd-trimester assessments. Women randomized to receive only usual prenatal care complete assessments on the same schedule as those in the intervention condition. We will use generalized estimating equation models and an intent-to-treat analysis to evaluate the efficacy of the intervention condition relative to the usual prenatal care condition in (1) increasing the proportion of women detected with a laboratory-confirmed negative PEth test during pregnancy and (2) reducing the proportion of adverse birth outcomes among infants. The proposed research is scientifically and clinically significant. If demonstrated to be effective, the intervention may represent a scalable alcohol reduction strategy suitable for prenatal clinical care in other maternity hospitals and clinical sites to reduce prenatal alcohol exposure and adverse birth outcomes. If successful, this would be the first prevention intervention in CIFASD.
This study develops, implements, and rigorously tests the effectiveness of a brief alcohol intervention in prenatal care. The objective of the alcohol intervention is to motivate women to reduce alcohol consumption during pregnancy. The study also assesses whether the intervention reduces adverse birth outcomes.
Assessment of Fetal Alcohol Spectrum Disorders (FASD) Using Novel Web-Based Tools
PI: Sarah Mattson
Assessment of Fetal Alcohol Spectrum Disorders (FASD) Using Novel Web-Based Tools U01 AA014834 | San Diego State University
 Our project is aimed at using online tools that can aid in the assessment and diagnosis of individuals with FASD. The two main tools used are the FASD-Tree, a screening tool for FASD, and the Brief Assessment of Individual Neurobehavior – online version (BRAIN-online), which assesses cognitive performance. Together, these tools will allow individual clinicians to assess patients for FASD in an efficient and cost-effective way. As part of the project, individuals who are being assessed for FASD will be examined using a physical exam, parent/caregiver questionnaires, and BRAIN-online. Results will be given to the clinician who will provide feedback to the individual and their family. In addition, patients will be assessed with measures from other CIFASD projects, including 3D facial imaging and other apps to detect facial features. Data from these projects will be combined with the ultimate goal of making diagnosis of FASD easier and cheaper to accomplish, especially in remote or less-resourced settings.
Our project is aimed at using online tools that can aid in the assessment and diagnosis of individuals with FASD. The two main tools used are the FASD-Tree, a screening tool for FASD, and the Brief Assessment of Individual Neurobehavior – online version (BRAIN-online), which assesses cognitive performance. Together, these tools will allow individual clinicians to assess patients for FASD in an efficient and cost-effective way. As part of the project, individuals who are being assessed for FASD will be examined using a physical exam, parent/caregiver questionnaires, and BRAIN-online. Results will be given to the clinician who will provide feedback to the individual and their family. In addition, patients will be assessed with measures from other CIFASD projects, including 3D facial imaging and other apps to detect facial features. Data from these projects will be combined with the ultimate goal of making diagnosis of FASD easier and cheaper to accomplish, especially in remote or less-resourced settings.
The detrimental effects of prenatal alcohol exposure, including cognitive, behavioral, and physical impacts, have been well documented over several decades. They include long-standing effects on multiple domains of function. Alcohol-affected individuals often go unrecognized. In addition, wait times for evaluation can be quite long, further delaying the accurate identification of affected individuals. Effective online screening tools can decrease wait times and streamline evaluations in specialty clinics. Prenatal alcohol exposure continues to represent a significant public health concern and increased efforts at documenting risk, improving identification, and providing clinically-efficient tools are paramount. As such, the overarching aim of this proposal is to improve the screening for prenatal alcohol exposure and fetal alcohol spectrum disorder (FASD) by deploying web-based tools that aid in clinical evaluation. Two main tools will be used, the FASD-Tree and the Brief Assessment of Individual Neurobehavior – online version (BRAIN-online). Both tools use web-based technology for collecting data and provide the clinician with information about the individual’s physical, behavioral, and cognitive attributes. Specifically, the FASD-Tree records physical and behavioral data, and BRAIN-online measures impulsivity, attention, problem-solving, processing speed, memory, spatial working memory, and set-shifting. To achieve the aims of this study, child subjects (6-17y) will be evaluated using either the combination of FASD-Tree and BRAIN-online or BRAIN-online alone (Aim 1-3). Parents will complete online questionnaires regarding their child’s background, demographics, and behavior and some children will be given a standardized in-person neuropsychological test battery. In Aim 1, we will examine the relation between FASD-Tree and BRAIN-online, including classification accuracy, sensitivity, and specificity for each tool and their combination. In Aim 2, data from these novel tools will be combined with traditional and advanced (e.g., MorpheusQ and 3D facial imaging) physical measurements collected through other CIFASD projects. The combination of measures used in the proposed research will result in a convenient, scalable, and cost-effective adjunct to standard diagnostic tools.
This project is directly relevant to public health concerns related to the effects of prenatal alcohol exposure on child health and development. This study aims to improve the screening of FASD by deploying web-based tools that aid in clinical evaluation. Two main tools will be used, the FASD-Tree and the Brief Assessment of Individual Neurobehavior – online version (BRAIN-online). Both tools use web-based technology for collecting data and provide the clinician with information about the individual’s physical, behavioral, and cognitive attributes. Data from these novel tools will be combined with traditional and advanced physical measurements collected through other CIFASD projects and will enhance clinical decision-making.
Leveraging Technology to Increase Quality of Life for FASD across the Lifespan
MPIs: Christie L. M. Petrenko and Cristiano Tapparello
Leveraging Technology to Increase Quality of Life for FASD across the Lifespan U01 AA026104 | University of Rochester

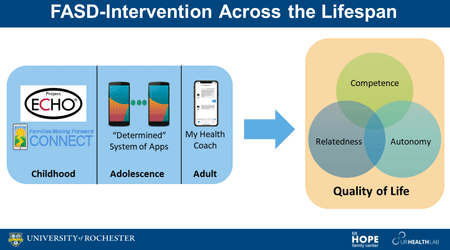
 Our goal is to improve quality of life for people with FASD across the lifespan. This project will test the effectiveness of phone and web applications to provide support at three different developmental periods. For children, we will train mental health providers to use our previously developed Families Moving Forward Connect app, using different methods to determine which is most effective. This app helps children and their families better cope with challenges faced. We are also developing apps for adolescents with FASD and their parents to support communication and skill building. Finally, we will test the My Health Coach app to help adults with FASD manage their health and daily life activities. These apps provide potential interventions that are highly accessible.
Our goal is to improve quality of life for people with FASD across the lifespan. This project will test the effectiveness of phone and web applications to provide support at three different developmental periods. For children, we will train mental health providers to use our previously developed Families Moving Forward Connect app, using different methods to determine which is most effective. This app helps children and their families better cope with challenges faced. We are also developing apps for adolescents with FASD and their parents to support communication and skill building. Finally, we will test the My Health Coach app to help adults with FASD manage their health and daily life activities. These apps provide potential interventions that are highly accessible.
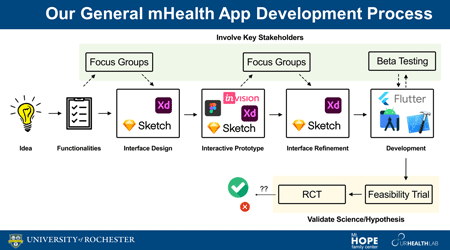
People with fetal alcohol spectrum disorders (FASD) experience barriers to care and a lower quality of life (QOL). Responsive to the Collaborative Initiative on FASD (CIFASD5) objectives of improving interventions and early case identification, this proposal evaluates three developmentally-appropriate and scalable interventions to improve QOL across the lifespan. Each intervention leverages technology to increase accessibility and overcome significant barriers to care. These technological interventions are versatile with good potential for dissemination, offering high potential public health impact. All three interventions are theoretically grounded in self-determination theory (SDT) and are integrated with useful best practices in “FASD-Informed Care,” derived from FASD research, clinical wisdom, and policy. Our methodological approach builds on our success developing mobile health (mHealth) applications within CIFASD4, which has included the Families Moving Forward (FMF) Connect app for caregivers of children ages 3-12 (U01 AA026104) and the My Health Coach app for adults with FASD (UH2 AA02050). Study aims will be accomplished using our established systematic user-centered design approach to mHealth interventions, which emphasizes engagement of key stakeholders throughout the development and testing process. Trial design and outcome measurement are guided by implementation science frameworks with the vision towards optimizing success of future dissemination in community settings. Aim 1 tests whether the “Provider-Assisted FMF Connect” intervention and an Extension of Community Healthcare Outcomes (ECHO) implementation package increases mental health providers’ (n=250) FASD-informed care knowledge, self- efficacy, and practice change (including screening and diagnosis of FASD). We hypothesize mental health providers trained in Provider-Assisted FMF Connect through ECHO tele-mentoring will evidence greater practice change compared to providers in self-directed implementation or waitlist conditions. A larger-scale efficacy trial, Aim 2 will test whether the My Health Coach app improves SDT and QOL outcomes for adults with FASD (n=120). Patterns of app usage relating to outcomes will guide further app refinements and dissemination. Leveraging advisory board and focus group input, Aim 3 will develop and assess usability of a caregiver-assisted mHealth app for adolescents called the “Determined” app system. The Determined app system will include both adolescent and caregiver apps, with synchronized features supporting adolescent self-determination skill building, caregiver autonomy-supportive parenting, and family QOL. Inclusion of these three aims across the lifespan facilitates efficient and mutually informative intervention development. It also addresses gaps in intervention research, especially in adolescence and adulthood. All three aims draw from diverse geographic regions, benefitting directly from recruitment via other CIFASD sites yet expanding beyond them. To other CIFASD5 projects and investigators, we provide much needed, scalable interventions to offer their participants and a clinical setting to test new diagnostic innovations. From findings of our CIFASD colleagues, we will glean content vital to improve our interventions.
Most people with fetal alcohol spectrum disorders (FASD) cannot access FASD-informed interventions and experience lower quality of life. This project will develop and evaluate three developmentally-appropriate FASD-informed interventions targeting quality of life at a different life phase. Each intervention leverages technology to increase accessibility and overcome significant barriers to care.
Defining Translational Approaches for the Image-Based Detection of Prenatal Alcohol Exposure
PI: Michael Suttie
Defining translational approaches for the image-based detection of prenatal alcohol exposure U01 AA014809 | University of Oxford
 Individuals with fetal alcohol spectrum disorder (FASD), which may have life-long impairments, including behavioral and learning difficulties. Thus, it is critical to improve diagnosis of FASD. To identify individuals with FASD, we rely on identifying specific facial features. While these features are recognizable in some individuals, they are often difficult to identify and can be very subtle in many of those affected, making diagnosis challenging. This project aims to apply machine learning (an application of artificial intelligence) to images, such as brain ultrasounds and 3D images of the face, and combine this information with data on an individual’s behaviors and learning abilities to identify those with FASD better. We are also exploring the how smoking and drug use in combination with alcohol may affect outcome. Ultimately, we are building and making tools available for clinicians to assist in the diagnosis of FASD.
Individuals with fetal alcohol spectrum disorder (FASD), which may have life-long impairments, including behavioral and learning difficulties. Thus, it is critical to improve diagnosis of FASD. To identify individuals with FASD, we rely on identifying specific facial features. While these features are recognizable in some individuals, they are often difficult to identify and can be very subtle in many of those affected, making diagnosis challenging. This project aims to apply machine learning (an application of artificial intelligence) to images, such as brain ultrasounds and 3D images of the face, and combine this information with data on an individual’s behaviors and learning abilities to identify those with FASD better. We are also exploring the how smoking and drug use in combination with alcohol may affect outcome. Ultimately, we are building and making tools available for clinicians to assist in the diagnosis of FASD.
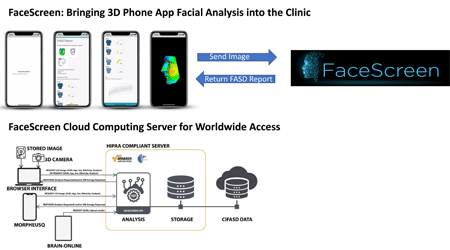
As a constituent component of CIFASD5, this project will focus on improving the identification of individuals with prenatal alcohol exposure (PAE), across the lifespan, with the primary goal of translation for clinical application. We target two main categories outlined in the Request for Application (RFA) 1. To identify FASD cases early and accurately, and 2. To expand and translate basic and mechanistic understandings of the effects of PAE. To address these topics, we aim to improve the accuracy and reliability of the clinical recognition of FASD, controlling for the influence of sex, co- exposures, and age. We seek to understand better how neurocognitive and facial morphology are correlative and covariant in FASD and exploit this relationship to improve screening accuracy. Previously, we have implemented methods and associated software for analyzing face shape and face – brain-cognitive relationships to detect the effects of PAE. In doing so, we have created an effective toolkit to automatically and objectively identify cardinal and subtle PAE-associated facial dysmorphism across the FASD spectrum. It is now a priority to refine and optimize our methods, utilizing low-cost 3D imaging devices to validate and distribute to the clinical community. To accomplish our goals, we propose to follow specific aims: 1. Explore the impact of factors secondary to alcohol exposure that may influence facial morphology; 2. Detect facial, cranial, and neural effects of prenatal alcohol exposure across the lifespan; 3. Develop, test, and validate automated methods for FASD screening, integrating neurocognitive assessment tools and distributing to the clinical community. Achieving aims 1 and 2 will dramatically refine FASD recognition by understanding the factors secondary to alcohol that may influence outcomes across the lifespan. Successful demonstration of aim 2 will achieve the earliest possible diagnosis enabling further research and feedback for intervention studies and also focus on recognizing traits in adults – a population significantly underreported in the literature. Ultimately, aim 3 encompasses the primary goal of the project, developing, testing, and validating multi-domain screening tools that assist with clinical identification. We will work closely with consortium partners who will recruit subjects and provide facial images and neurocognitive assessment data (U01: Chambers; U01: Coles & Weinberg; U01: Wozniak; U01: DiClemente; U01: Petrenko). Particularly close collaboration is required for the face-neurocognitive analysis, where we will integrate and combine facial and neurocognitive screening tools (U01: Mattson). We will rely on research resources for data management (R24: Wetherill) and to validate screening tools and gain clinical feedback (R24: Del Campo).
Automated tools will be developed to better identify those with suspected prenatal alcohol exposure using a combination of 3D facial imaging and neurocognitive testing using low-cost imaging devices. We will utilize outputs from online cognitive assessment (FASD-Tree and Brain-Online) tools to improve sensitivity/specificity for the identification of FASD. Machine learning approaches will be applied to achieve automated objective analysis of facial images and deployed on a cloud server for worldwide, multi-device accessibility. To ensure our tools are optimized, we will investigate the impact on facial dysmorphism of age, ethnicity, sexual dimorphism, and co-exposures. These tools will facilitate and support the recognition of individuals with FASD, who may need further support, and will include infants through to adults using a fully automated and objective approach, using low-cost solutions and providing an accessible toolkit for clinicians.
tDCS and Cognitive Training as a Neurodevelopmental Intervention in FASD
PI: Jeff Wozniak
tDCS and Cognitive Training as a Neurodevelopmental Intervention in FASD U01 AA030164 | University of Minnesota
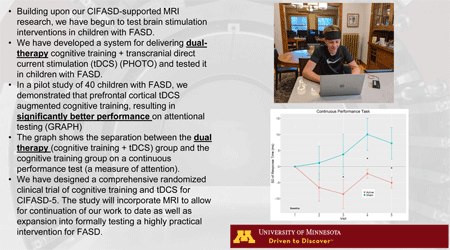
 Prenatal alcohol exposure can lead to brain abnormalities and brain dysfunction that affects individuals’ cognitive abilities. This project will refine and test an intervention that targets brain activity, mild brain stimulation delivered to the scalp, combined with computerized cognitive training. Our goal is to determine if this intervention improves attentional functioning and impulse control in children and adolescents with FASD. If effective, this method could be readily scaled and applied widely as a treatment for a population that currently has few such options.
Prenatal alcohol exposure can lead to brain abnormalities and brain dysfunction that affects individuals’ cognitive abilities. This project will refine and test an intervention that targets brain activity, mild brain stimulation delivered to the scalp, combined with computerized cognitive training. Our goal is to determine if this intervention improves attentional functioning and impulse control in children and adolescents with FASD. If effective, this method could be readily scaled and applied widely as a treatment for a population that currently has few such options.
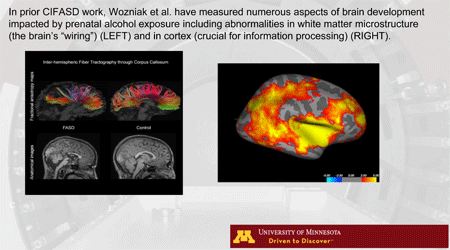
Fetal alcohol spectrum disorders (FASDs) comprise a range of effects from prenatal alcohol exposure (PAE) including neurological abnormalities, cognitive and behavioral impairments, growth retardation, and craniofacial anomalies. Very few treatments have been investigated despite the tremendous public health burden posed by FASD. Cognition is a natural target for intervention because deficits contribute to problems with adaptive functioning, social skills, and independent living. A decade of research suggests that cognitive training has beneficial effects on functional brain networks – mediating cognition in a variety of neurodevelopmental and mental health conditions. We believe it can improve cognitive performance in individuals with FASD. The primary objective of our existing line of neuromodulation research and the proposed RCT is to leverage the ability of tDCS to enhance cortical excitability and neural plasticity in order to maximize the cognitive training benefits in children and adolescents with FASD. Recently we carried out a pilot randomized controlled trial of tDCS-augmented cognitive training to demonstrate the safety and feasibility of this approach in children with FASD (Boroda, Krueger, et al., 2020). We showed that the intervention was safe and well-tolerated in this population. Further, despite applying a relatively low-dose of tDCS and cognitive training, we found that the group receiving active-tDCS demonstrated greater improvements on an attention task than a group receiving sham-tDCS. These promising pilot results merit replication in a larger clinical trial. We propose a fully-powered randomized controlled trial of 60 (at completion) children and adolescents with FASD who will receive cognitive training in combination with tDCS. We hypothesize that, in conjunction with cognitive training, five initial sessions of active tDCS will produce a significantly greater improvement in sustained attention (CPT) than sham tDCS. We also hypothesize that, in conjunction with cognitive training, a total of ten sessions of active tDCS will produce a significantly greater improvement in sustained attention (CPT) than five sessions of active tDCS. We also aim to establish the long-term durability of neurocognitive improvements and associated behavioral changes resulting from tDCS and cognitive training. Finally, we plan to use pre-intervention and post-intervention resting-state fMRI data to show functional network changes related to the intervention and to quantify the relationship between dose and functional network changes. Results of the trial will directly inform future clinical implementation of cognitive training and tDCS as a potential neurodevelopmental intervention.
We have conducted a pilot study of an intervention for children with neurodevelopmental abnormalities resulting from prenatal alcohol exposure (PAE) that consists of a combination of computerized cognitive training (attention and executive functioning) and strategic brain stimulation (transcranial direct current stimulation (tDCS)) – which is very low voltage stimulation designed to momentarily enhance “plasticity” in the cortex. The study will test five initial sessions of cognitive training and active tDCS vs. sham tDCS (placebo) and then go on to test two different dosages (5 active sessions vs. 10 active sessions). Outcome measures will assess attention, executive functioning, parent-reported behaviors related to these underlying cognitive functions, and functional brain network changes using state-of-the-art functional MRI methodologies.
Lifelong Impact of PAE on Stem Cell Dynamics and Cellular Aging
PI: Amanda Mahnke
Lifelong Impact of PAE on Stem Cell Dynamics and Cellular Aging< UH2 AA030186 | Texas A&M University Health Science Center
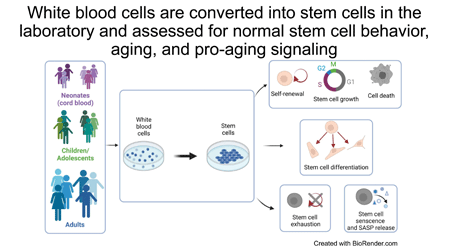
 Stem cells support healthy aging, creating cells to repair and regenerate tissues. But stem cells also contribute to the negative consequences of aging, becoming exhausted, losing repair capabilities, and releasing signals that encourage other stem cells to stop replicating. Research indicates that aging is different for individuals with FASD, including earlier onset of some aging-associated diseases. We will use stem cells created in the laboratory to determine how stem cell repair and regeneration ability is altered for individuals with FASD and if stem cells are prematurely aging and releasing more pro-aging signals. Our goal is to use these data to identify ways to therapeutically address changes in stem cell behavior and support healthy aging for individuals with FASD.
Stem cells support healthy aging, creating cells to repair and regenerate tissues. But stem cells also contribute to the negative consequences of aging, becoming exhausted, losing repair capabilities, and releasing signals that encourage other stem cells to stop replicating. Research indicates that aging is different for individuals with FASD, including earlier onset of some aging-associated diseases. We will use stem cells created in the laboratory to determine how stem cell repair and regeneration ability is altered for individuals with FASD and if stem cells are prematurely aging and releasing more pro-aging signals. Our goal is to use these data to identify ways to therapeutically address changes in stem cell behavior and support healthy aging for individuals with FASD.
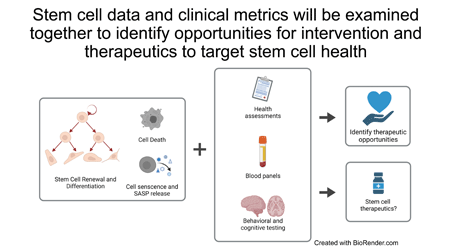
Prenatal alcohol exposure (PAE) is common and can result in brain-based disabilities and growth deficits. The impact of PAE is not just in early development, but also can lead to secondary health problems throughout the lifespan. These secondary problems can include higher rate and earlier onset of aging-related diseases, including cardiovascular disease, autoimmune disorders such as arthritis, and decreased bone density. The early onset of these aging-related diseases indicates that a consequence of PAE is premature aging of tissues and organs. There is an unmet need to better understand this PAE-induced premature aging and determine the underlying mechanisms that could be leveraged to delay or prevent these secondary health conditions. We know that PAE is a potent teratogen that reprograms stem cells. Our hypothesis is that this stem cell reprogramming has lifelong consequences, including the premature aging of stem cells as a mechanism that drives systemic aging. This hypothesis is supported by published literature that shows PAE can disrupt stem cell self-renewal, due, in part, to premature or aberrant differentiation, and that these disrupted stem cell behaviors persist into adulthood. Based on these data, we plan to address two questions: firstly, “does PAE in human populations diminish stem cell function across the lifespan?”; secondly, “does PAE induce or exacerbate human stem cell aging?”. To address the above two questions, we plan to create human induced pluripotent stem cells (hiPSCs), as early passages of these cells retain epigenetic markers of aging. These cells will be derived from regionally and ethnically diverse neonatal, child/adolescent, and adult cohorts of individuals with PAE/fetal alcohol spectrum disorders (FASDs) and from matched controls. In Aim 1 we plan to use a panel of cellular and molecular assays to assess PAE/FASD-induced changes in stem cell growth, self-renewal, and trilineage (ectoderm, mesoderm, endoderm) differentiation. In Aim 2 we plan to assess alterations to stem cell aging, including exhaustion, senescence, and release of pro-inflammatory molecules as part of the senescence- associated secretory phenotype. Our overarching goal, to ultimately identify underlying mechanisms mediate the emergence of secondary health conditions for individuals with FASDs, is consistent with the mission of the NIAAA (RFA-AA-21-014). PAE is known to inhibit stem cell function. However, tissue stem cells may also be a novel target for the prevention and treatment of PAE-induced premature aging. At the conclusion of these studies, we will have: firstly, created a unique community resource, a panel of patient-derived hiPSC cells, that can be used to assess the systemic impact of PAE across the lifespan; secondly, expanded our knowledge of the impact of PAE on stem cell behavior; and thirdly, identified important cellular mechanisms of premature aging.
Prenatal alcohol exposure (PAE) is the leading cause of neurodevelopmental disability in the US and impacts health across the lifespan, with higher rate and earlier onset of aging-associated conditions. However, the mechanisms underlying the premature onset of these aging-associated conditions are unknown, making it imperative to determine how PAE impacts the body across aging and to identify windows for intervention to prevent these secondary conditions. The goals of this project are relevant to public health and consistent with the goals of RFA-AA-21-014 to ”translate basic and mechanistic understandings of the effects of PAE” and to “improve interventions to mitigate FASD outcomes”.
Development of biomarkers in deciduous teeth of children with FASD that predict neurobehavioral performance
MPIs: Annika Montag and Christine Austin
Development of biomarkers in deciduous teeth of children with FASD that predict neurobehavioral performance 1UH2AA029062-01 | University of California San Diego

 Prenatal alcohol exposure sequelae, collectively termed Fetal Alcohol Spectrum Disorders (FASD), are varied and present in as many as 10% of elementary school children. They affect cognitive functioning, as well as physical and mental health, and represent a significant public health issue. Timely and appropriate treatment can positively impact a child’s developmental trajectory and prevent secondary disabilities. In the majority of cases, diagnosis of FASD requires documentation of prenatal alcohol exposure. A biomarker of prenatal alcohol exposure would allow diagnosis of children where maternal self-report is unavailable. Prevention and treatment efforts would benefit from biomarkers able to noninvasively assess the magnitude and gestational time of exposures. Additionally, research into the etiology of FASD would benefit from such markers and the refining of our current understanding of mechanisms. We propose to develop biomarkers in dental tissue to quantitatively measure exposures, allowing the documentation of prenatal alcohol exposure in naturally shed deciduous (baby) teeth and the linking of prenatal exposures to neurobehavioral deficits. We will optimize our techniques to be able to detect exposures by month of second and third trimester. We will test whether our novel biomarkers predict neurobehavioral performance. In teasing out associations among exposures and outcomes, our study benefits from the well-characterized CIFASD cohort where consistently gathered, already existing data may be accessed and analyzed in conjunction with novel biomarker findings. This study will provide preliminary data for an R01 or U01 application to incorporate additional outcome data existing within the array of CIFASD data, such as neuroimaging, assess the effects of co-exposures, and further calibrate and improve our predictive capacity within the larger sample.
Prenatal alcohol exposure sequelae, collectively termed Fetal Alcohol Spectrum Disorders (FASD), are varied and present in as many as 10% of elementary school children. They affect cognitive functioning, as well as physical and mental health, and represent a significant public health issue. Timely and appropriate treatment can positively impact a child’s developmental trajectory and prevent secondary disabilities. In the majority of cases, diagnosis of FASD requires documentation of prenatal alcohol exposure. A biomarker of prenatal alcohol exposure would allow diagnosis of children where maternal self-report is unavailable. Prevention and treatment efforts would benefit from biomarkers able to noninvasively assess the magnitude and gestational time of exposures. Additionally, research into the etiology of FASD would benefit from such markers and the refining of our current understanding of mechanisms. We propose to develop biomarkers in dental tissue to quantitatively measure exposures, allowing the documentation of prenatal alcohol exposure in naturally shed deciduous (baby) teeth and the linking of prenatal exposures to neurobehavioral deficits. We will optimize our techniques to be able to detect exposures by month of second and third trimester. We will test whether our novel biomarkers predict neurobehavioral performance. In teasing out associations among exposures and outcomes, our study benefits from the well-characterized CIFASD cohort where consistently gathered, already existing data may be accessed and analyzed in conjunction with novel biomarker findings. This study will provide preliminary data for an R01 or U01 application to incorporate additional outcome data existing within the array of CIFASD data, such as neuroimaging, assess the effects of co-exposures, and further calibrate and improve our predictive capacity within the larger sample.
This study will determine whether prenatal alcohol exposure can be detected and quantified by gestational timing in naturally shed deciduous teeth of children with FASD who are enrolled in the CIFASD consortium. The study will further determine if timing and magnitude of prenatal alcohol exposure predict neurobehavioral outcomes assessed in 5-12-year-old children with FASD. This work will provide insights into FASD etiology and mechanisms, and support more precise delineation of diagnostic categories.
Mobile Health Tools to Promote Health in Adults With FASD
MPIs: Christie L. M. Petrenko and Cristiano Tapparello
Mobile Health Tools to Promote Health in Adults With FASD 1UH2AA029050-01 | University of Rochester

 Adults with fetal alcohol spectrum disorders (FASD) experience many barriers to care. Research on the effects of prenatal alcohol exposure during adulthood is limited and few providers have the knowledge and skills to effectively serve this population. Adults with FASD need reliable and accessible information to inform decision making about their health and well-being. Innovative and scalable solutions are needed. The proposed project aims to meet this need by developing a novel mobile health (mHealth) application (“app”), currently called “My Health Coach,” to directly provide adults with FASD evidence-based education about their condition and tools to promote their own self-management and health advocacy goals. We will build on our success with the Families Moving Forward (FMF) Connect mHealth app for caregivers of younger children with FASD. This will allow us to extend into adulthood and expedite our ability to innovate adaptations tailored for this population. This project will follow a systematic approach to the development and evaluation of the My Health Coach app. We will engage an advisory board of adult leaders with FASD and seek input from key stakeholders at each step of the development and testing process. We will utilize focus group and survey methodologies to solicit input from diverse adults to inform development of the My Health Coach app (Aim 1). We will then conduct a feasibility trial (n=40; Aim 2), including both qualitative and quantitative methodologies to assess the feasibility and user satisfaction and experiences with the app. The results will inform further refinements of the app and estimate effect sizes for future larger-scale trials. Implementation data will also aid in identifying the patterns of app usage that relate to the greatest improvements in outcomes. Study hypotheses include: (1) adults with FASD will appreciate the simple and engaging design of the app, its ability to organize and remind them of information, and its delivery of useful content when they need it; (2) participants will vary in their perceived motivators and barriers for app use; and (3) improvements in pre-post outcomes will be associated with a usage pattern reflecting more routine app usage and a higher frequency of user interactions with the app. Results of this study will further the overall strategic aims of the Collaborative Initiative on FASD (CIFASD), which are to inform and develop effective interventions for FASD. This project integrates two main themes of CIFASD-4, specifically (1) adult health, and (2) using technology to increase access to care. The project will also benefit from resources and collaborations within CIFASD to carry out the proposed work, including integrating findings from other projects into inform intervention content, recruitment of a diverse sample, and outreach and dissemination. This will be the first study to systematically develop and test an intervention for adults with FASD targeting health and well-being. It has the advantage of being scalable and could reduce significant barriers to care, resulting in a greater public health impact.
Adults with fetal alcohol spectrum disorders (FASD) experience many barriers to care. Research on the effects of prenatal alcohol exposure during adulthood is limited and few providers have the knowledge and skills to effectively serve this population. Adults with FASD need reliable and accessible information to inform decision making about their health and well-being. Innovative and scalable solutions are needed. The proposed project aims to meet this need by developing a novel mobile health (mHealth) application (“app”), currently called “My Health Coach,” to directly provide adults with FASD evidence-based education about their condition and tools to promote their own self-management and health advocacy goals. We will build on our success with the Families Moving Forward (FMF) Connect mHealth app for caregivers of younger children with FASD. This will allow us to extend into adulthood and expedite our ability to innovate adaptations tailored for this population. This project will follow a systematic approach to the development and evaluation of the My Health Coach app. We will engage an advisory board of adult leaders with FASD and seek input from key stakeholders at each step of the development and testing process. We will utilize focus group and survey methodologies to solicit input from diverse adults to inform development of the My Health Coach app (Aim 1). We will then conduct a feasibility trial (n=40; Aim 2), including both qualitative and quantitative methodologies to assess the feasibility and user satisfaction and experiences with the app. The results will inform further refinements of the app and estimate effect sizes for future larger-scale trials. Implementation data will also aid in identifying the patterns of app usage that relate to the greatest improvements in outcomes. Study hypotheses include: (1) adults with FASD will appreciate the simple and engaging design of the app, its ability to organize and remind them of information, and its delivery of useful content when they need it; (2) participants will vary in their perceived motivators and barriers for app use; and (3) improvements in pre-post outcomes will be associated with a usage pattern reflecting more routine app usage and a higher frequency of user interactions with the app. Results of this study will further the overall strategic aims of the Collaborative Initiative on FASD (CIFASD), which are to inform and develop effective interventions for FASD. This project integrates two main themes of CIFASD-4, specifically (1) adult health, and (2) using technology to increase access to care. The project will also benefit from resources and collaborations within CIFASD to carry out the proposed work, including integrating findings from other projects into inform intervention content, recruitment of a diverse sample, and outreach and dissemination. This will be the first study to systematically develop and test an intervention for adults with FASD targeting health and well-being. It has the advantage of being scalable and could reduce significant barriers to care, resulting in a greater public health impact.
Adults with fetal alcohol spectrum disorders (FASD) experience many barriers to care and have difficulty accessing reliable information about their condition. This project will develop and evaluate the efficacy of a mobile health application to support their health and well-being.
Choline Polymorphisms in FASD
Susan M. Smith
Choline Polymorphisms in FASD 1UH2AA029056-01 | University of North Carolina Chapel Hill
 This exploratory/developmental UH2 pilot application to CIFASD addresses the need for improved intervention in Fetal Alcohol Spectrum Disorder (FASD), a leading cause of life-long behavioral and cognitive disability. This proposal focuses on the nutrient choline, a one-carbon donor that is essential for healthy brain development. Polymorphisms in choline-metabolizing genes affect its synthesis, transport, and utilization, and thus affect choline need and efficacy of its use. Strong preclinical data show that choline supplementation – both in utero and postnatally – mitigates the cognitive deficits due to prenatal alcohol exposure (PAE). Clinical studies have more nuanced outcomes, and choline supplements confer more modest or even no benefit; however, variables including age, developmental brain stage, and duration confound the interpretation. Our recent SNP analysis of subjects in the Wozniak intervention found that polymorphisms in the choline transporter SLC44A1 (CTL1) predict who benefited most from choline; specifically, subjects having minor alleles in SLC44A1 have the greatest memory improvement when given supplemental choline. SLC44A1 is ubiquitous and its activity is reduced by low choline intake, and these minor alleles further reduce its activity. Thus, those with the minor alleles are the most vulnerable to choline inadequacy and benefit most from its supplementation. Here, we collaborate with CIFASD investigators to investigate the role of SLC44A1 in FASD. Specifically, we hypothesize that polymorphisms in SLC44A1 significantly influence behavioral outcomes in FASD, in both the presence and absence of choline intervention. Aim 1 tests the hypothesis that, within the Ukrainian intervention trial, those PAE pregnancies with minor alleles in SLC44A1 derive the greatest cognitive benefit from choline supplementation. Aim 2 tests the hypothesis that, of individuals diagnosed with FASD and not receiving choline, those having minor alleles in SLC44A1 will have the poorest cognitive performance. Aim 3 expresses these minor allelic proteins in a human neuronal lineage, to understand the functional consequence of these variants to choline transport and metabolism. Aims 1-2 utilize the CIFASD database, in a collaboration with CIFASD investigators Christina Chambers, Tatiana Foroud and Jeffrey Wozniak, and with choline expert Steven Zeisel. These findings (i) identify who benefits most from choline intervention; (ii) informs how choline improves outcomes in FASD; and (iii) enables optimization of the choline intervention. This study represents the first application of Personalized Medicine to FASD. The results position us to join a CIFASD U01 that would validate SLC44A1’s influence in an independent cohort of gestational substance abuse, with testing for additional choline-related polymorphisms that further influence response to choline intervention.
This exploratory/developmental UH2 pilot application to CIFASD addresses the need for improved intervention in Fetal Alcohol Spectrum Disorder (FASD), a leading cause of life-long behavioral and cognitive disability. This proposal focuses on the nutrient choline, a one-carbon donor that is essential for healthy brain development. Polymorphisms in choline-metabolizing genes affect its synthesis, transport, and utilization, and thus affect choline need and efficacy of its use. Strong preclinical data show that choline supplementation – both in utero and postnatally – mitigates the cognitive deficits due to prenatal alcohol exposure (PAE). Clinical studies have more nuanced outcomes, and choline supplements confer more modest or even no benefit; however, variables including age, developmental brain stage, and duration confound the interpretation. Our recent SNP analysis of subjects in the Wozniak intervention found that polymorphisms in the choline transporter SLC44A1 (CTL1) predict who benefited most from choline; specifically, subjects having minor alleles in SLC44A1 have the greatest memory improvement when given supplemental choline. SLC44A1 is ubiquitous and its activity is reduced by low choline intake, and these minor alleles further reduce its activity. Thus, those with the minor alleles are the most vulnerable to choline inadequacy and benefit most from its supplementation. Here, we collaborate with CIFASD investigators to investigate the role of SLC44A1 in FASD. Specifically, we hypothesize that polymorphisms in SLC44A1 significantly influence behavioral outcomes in FASD, in both the presence and absence of choline intervention. Aim 1 tests the hypothesis that, within the Ukrainian intervention trial, those PAE pregnancies with minor alleles in SLC44A1 derive the greatest cognitive benefit from choline supplementation. Aim 2 tests the hypothesis that, of individuals diagnosed with FASD and not receiving choline, those having minor alleles in SLC44A1 will have the poorest cognitive performance. Aim 3 expresses these minor allelic proteins in a human neuronal lineage, to understand the functional consequence of these variants to choline transport and metabolism. Aims 1-2 utilize the CIFASD database, in a collaboration with CIFASD investigators Christina Chambers, Tatiana Foroud and Jeffrey Wozniak, and with choline expert Steven Zeisel. These findings (i) identify who benefits most from choline intervention; (ii) informs how choline improves outcomes in FASD; and (iii) enables optimization of the choline intervention. This study represents the first application of Personalized Medicine to FASD. The results position us to join a CIFASD U01 that would validate SLC44A1’s influence in an independent cohort of gestational substance abuse, with testing for additional choline-related polymorphisms that further influence response to choline intervention.
Intervention trials of the dietary nutrient choline show inconsistent benefits in improving behavioral outcomes in Fetal Alcohol Spectrum Disorder (FASD), a leading cause of preventable neurobehavioral disability. This proposal tests the hypothesis that polymorphisms in choline-related genes significantly modulate the efficacy of supplemental choline to improve behavioral outcomes in alcohol-exposed pregnancies, and in children with FASD. Findings from this work enable us to optimize the choline interventions in alcohol-exposed pregnancies, as well as those diagnosed with FASD.